A Mysterious Mineral on Mars May Rewrite the Planet’s Geologic History

A potential new mineral on Mars forms when iron sulfates are heated above 100°C. Data from Valles Marineris regions suggest geothermal processes altered ancient sulfate deposits.
A study published in Nature Communications reports the discovery of an iron sulfate on Mars that may represent a previously unknown mineral. Sulfur is abundant on the planet and often combines with other elements to create sulfate minerals.
On Earth, most sulfates dissolve easily when exposed to rain. Mars, however, has an extremely dry surface. Because of this, sulfate minerals can remain stable for billions of years, preserving valuable evidence about the planet’s early conditions. Every mineral has its own crystal structure and physical properties, including well-known examples such as gypsum and hematite.
Researchers use measurements from spacecraft orbiting Mars to detect minerals on the planet’s surface and to reconstruct the environments where those minerals formed. For nearly two decades, scientists have been trying to explain unusual layered iron sulfates that show a distinctive spectral signal in orbital data.
Solving a 20-Year Mystery of Martian Iron Sulfates
A research team led by Dr. Janice Bishop, senior research scientist at the SETI Institute and NASA’s Ames Research Center in California’s Silicon Valley, combined laboratory experiments with satellite observations of Mars. Their work identified and described a rare ferric hydroxysulfate phase. The findings provide new understanding of how heat, water, and chemical processes have shaped the Martian surface.
“We investigated two sulfate-bearing sites near the vast Valles Marineris canyon system that included mysterious spectral bands seen from orbital data, as well as layered sulfates and intriguing geology,” said Bishop.
The investigation focused on two locations. One was Aram Chaos, northeast of Valles Marineris, where ancient water once flowed toward lower northern regions. The other was the plateau above Juventae Chasma, a canyon about 5 kilometers deep located just north of Valles Marineris.
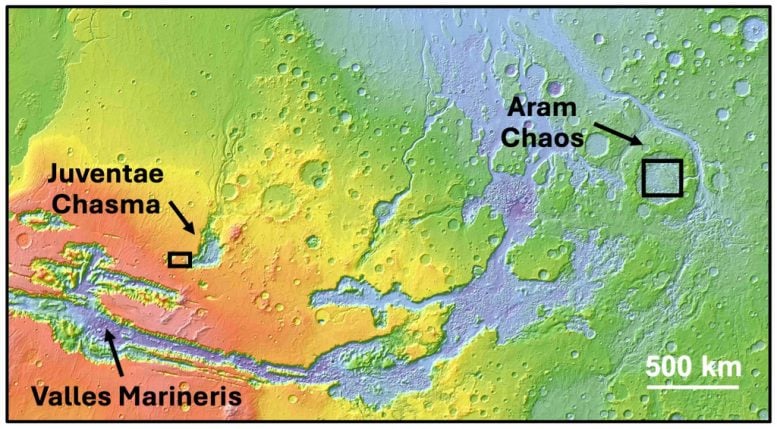
This region near the cliffs of Valles Marineris preserves signs of a wetter past. Ancient water channels are visible across the terrain. Yet sulfates appear concentrated in a small, low-lying area. Scientists think these deposits formed when pools of sulfate rich water slowly evaporated, leaving behind hydrated ferrous sulfates. These minerals, including ferric hydroxysulfate, occur as thin layers roughly a meter thick both above and below basaltic rocks. This layering indicates that the deposits were heated by lava or volcanic ash after they formed.
Evidence of Ancient Water and Layered Sulfate Deposits
“Investigation of the morphologies and stratigraphies of these four compositional units allowed us to determine the age and formation relationships among the different units,” said Dr. Catherine Weitz, a co-author on the study and senior scientist at the Planetary Science Institute.
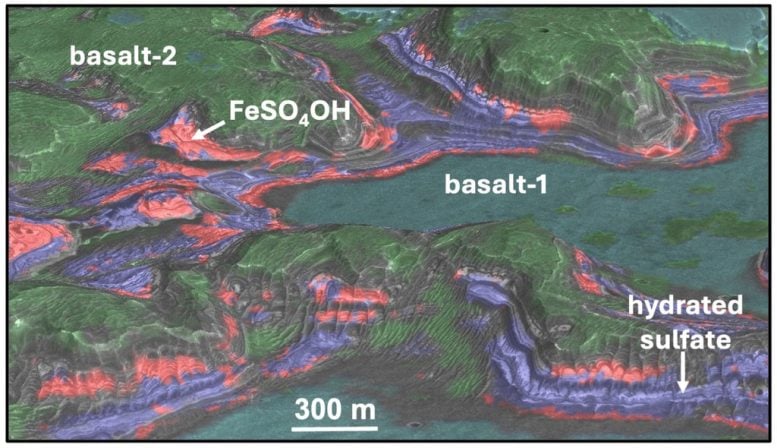
Sulfate minerals appear throughout the Valles Marineris region, including within rugged areas known as chaotic terrains. Scientists believe these landscapes formed when powerful floods reshaped the surface long ago. As the water disappeared, it left layered deposits of iron and magnesium sulfates that serve as important evidence of Mars’s wetter past. In one chaotic terrain that developed inside an ancient impact crater, the top layers contain polyhydrated sulfates, while deeper layers contain monohydrated sulfates and ferric hydroxysulfate.

Each of these sulfate minerals produces a unique spectral signature that spacecraft instruments can detect. Researchers used the CRISM instrument to identify them from orbit. The order of the layers was initially difficult to explain. Laboratory experiments revealed that heating polyhydrated sulfates to about 50°C creates monohydrated forms, while temperatures above 100°C generate ferric hydroxysulfate. These results suggest that geothermal heat likely drove the mineral transformations.
Mapping Sulfate Distribution and Geothermal Activity
Polyhydrated and monohydrated sulfates are widespread across the region. Ferric hydroxysulfate, in contrast, appears only in a few limited areas. The hottest geothermal sources likely existed beneath the locations where ferric hydroxysulfate is found today. Additional deposits could remain hidden beneath layers of monohydrated sulfates.
To better understand these changes, scientists at the SETI Institute and NASA Ames conducted laboratory experiments that reproduced the transformation of sulfate minerals. The sequence begins with rozenite (Fe²⁺SO₄·4H₂O), which contains four water molecules per unit cell. Heating converts it into szomolnokite (Fe²⁺SO₄·H₂O), which contains one water molecule. Further heating produces ferric hydroxysulfate, where OH replaces H₂O in the mineral structure.
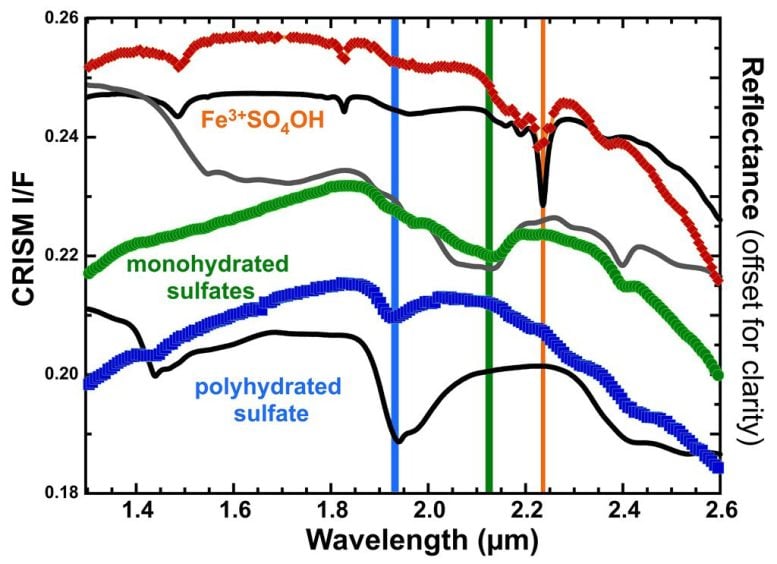
“Our experiments suggest that this ferric hydroxysulfate only forms when hydrated ferrous sulfates are heated in the presence of oxygen,” said postdoctoral researcher Dr. Johannes Meusburger at NASA Ames. “While the changes in the atomic structure are very small, this reaction drastically alters the way these minerals absorb infrared light, which allowed identification of this new mineral on Mars using CRISM.”
This chemical reaction requires oxygen gas and releases water (Equation 1). Mars currently has a thin atmosphere composed mostly of CO₂, but it still contains enough oxygen for this reaction to occur and for iron minerals to oxidize.
Equation 1: 4 Fe2+SO4·H2O + O2 → 4 Fe3+SO4OH + 2H2O
Laboratory Evidence for a Potentially New Mineral
“The material formed in these lab experiments is likely a new mineral due to its unique crystal structure and thermal stability,” said Bishop. “However, scientists must also find it on Earth to officially recognize it as a new mineral.”
The newly identified ferric hydroxysulfate has a structure similar to szomolnokite, which is a monohydrated ferrous sulfate mineral. However, it appears to form more readily from rozenite, a tetrahydrated sulfate mineral.
The conversion from hydrated ferrous sulfates to ferric hydroxysulfate requires temperatures above 100°C, which is far warmer than typical surface temperatures on Mars. Scientists believe the sulfates at Aram Chaos and Juventae, including ferric hydroxysulfate, formed later than the surrounding terrain, possibly during the Amazonian period (<3 billion years ago).
The study indicates that heat from volcanic activity at the Juventae Plateau and geothermal energy beneath Aram Chaos could transform common hydrated sulfates into ferric hydroxysulfate. These results suggest that parts of Mars remained chemically and thermally active more recently than previously thought. The findings provide new insight into the planet’s evolving surface and its past potential to support life.
Reference: “Characterization of ferric hydroxysulfate on Mars and implications of the geochemical environment supporting its formation” by J. L. Bishop, J. M. Meusburger, C. M. Weitz, M. Parente, C. Gross, D. Talla, A. M. Saranathan, Y. Itoh, M. R. D. Gruendler, A. E. G. Howells, M. Yeşilbaş, T. Hiroi, B. Schmitt, A. Maturilli, M. Al-Samir, T. F. Bristow, B. Lafuente and M. Wildner, 5 August 2025, Nature Communications.
DOI: 10.1038/s41467-025-61801-2
The paper, Characterization of Ferric Hydroxysulfate on Mars and Implications of the Geochemical Environment Supporting its Formation, is published in Nature Communications.
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.
Source link

