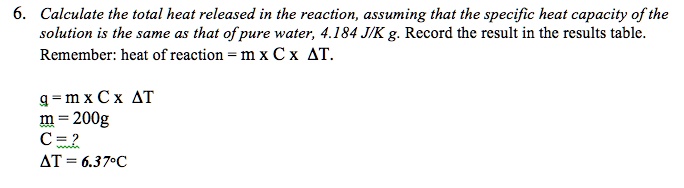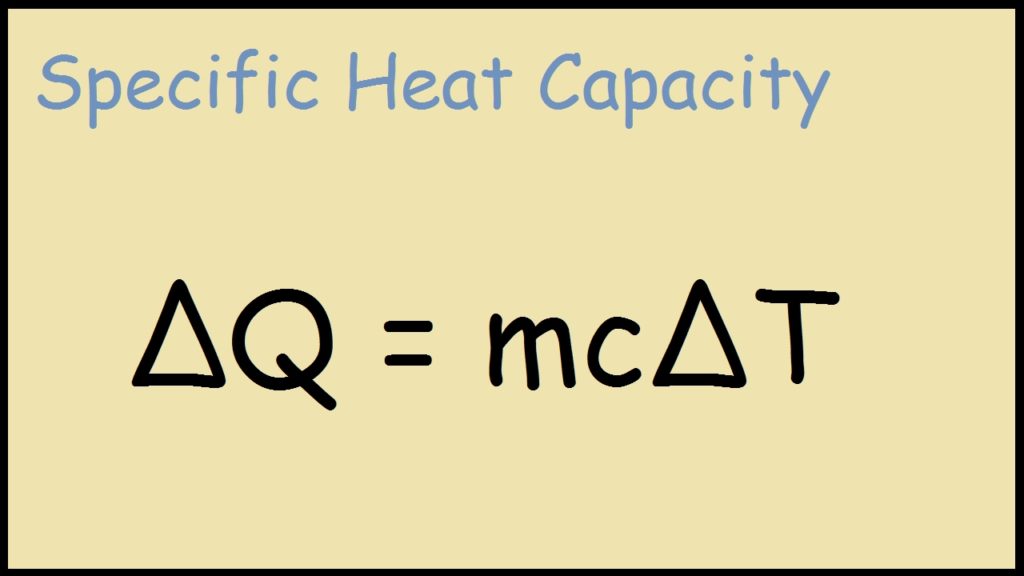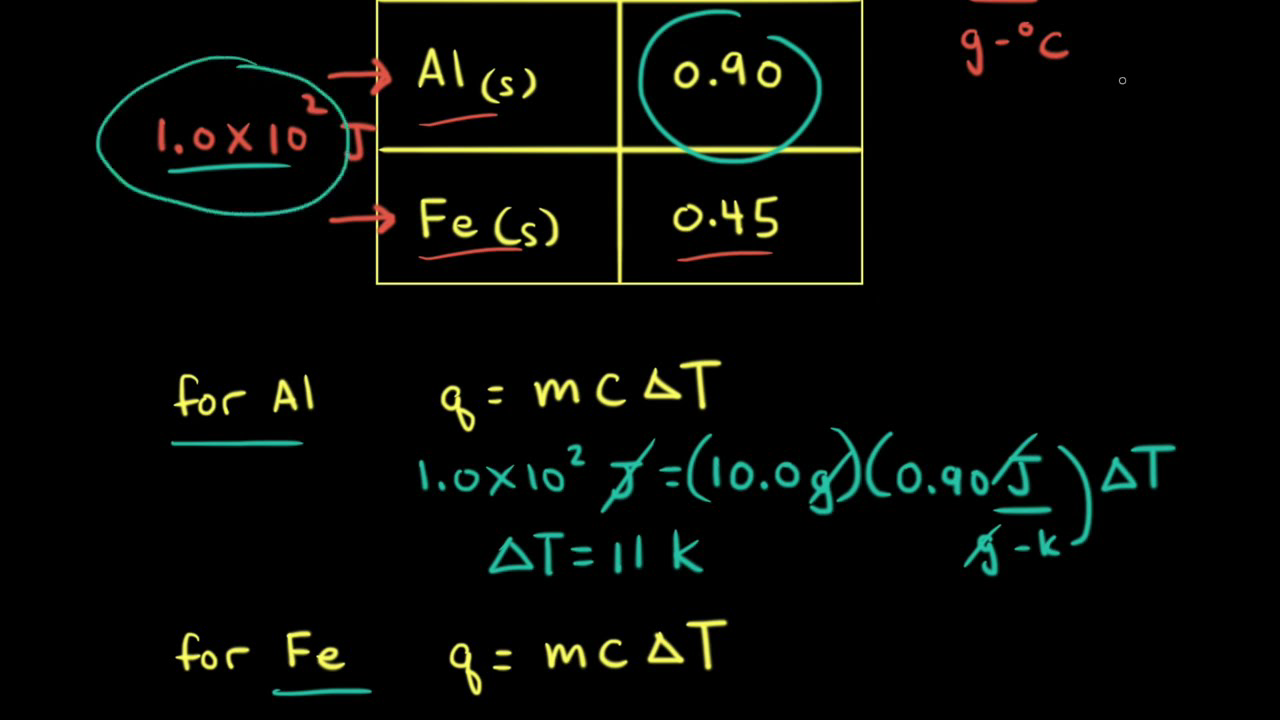How to determine the heat evolved or consumed by the reaction of 1.0 g SO2(g) with excess oxygen, with results from a Hess' Law Equation? | Socratic

Question Video: Calculating the Heat Energy Transferred to Water Using Its Specific Heat Capacity | Nagwa

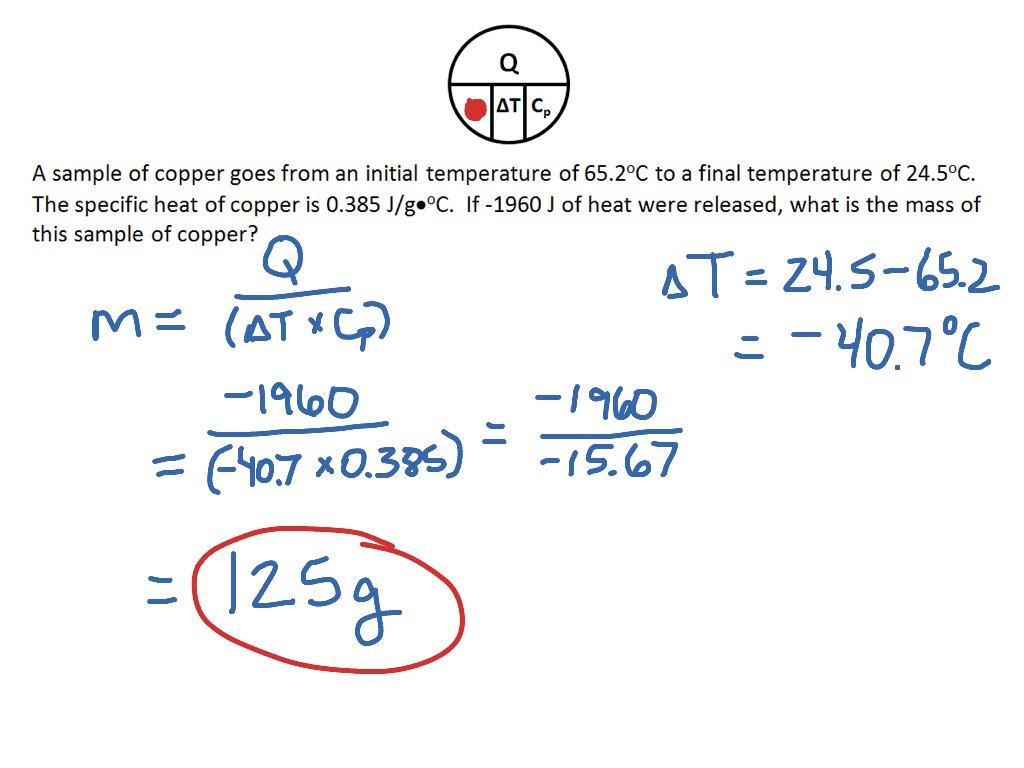
Heat Capacity and Specific Heat Calculations - Practice - A 1 g sample of decane, C 10 H 22 , is - Studocu