Scientists Say Conquering Age-Related Diseases Could Dramatically Extend Human Life

A new analysis suggests many anti-aging claims may reflect disease-specific improvements rather than genuine slowing of aging.
The analysis, published in Genomic Psychiatry, calls on researchers to rethink how biological aging is measured and interpreted. Dr. Dan Ehninger, who leads the Translational Biogerontology Laboratory at the German Center for Neurodegenerative Diseases, and Dr. Maryam Keshavarz conducted a systematic review examining widely used indicators of aging.
Their work argues that common measures such as lifespan extension, epigenetic clocks, frailty indices, and even the widely cited hallmarks of aging framework may blur the line between true changes in aging and general physiological effects that occur regardless of age.
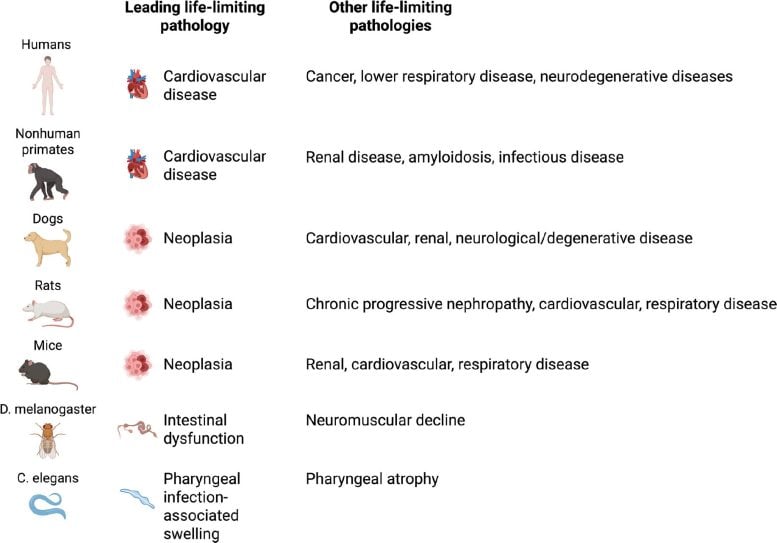
One of the most surprising insights comes from comparing causes of death across species. In humans, cardiovascular disease is responsible for roughly 35 to 70 percent of deaths among older adults. Autopsy studies prove that even centenarians who appeared healthy shortly before death almost always died from identifiable medical conditions rather than from old age alone.
Research on people between 97 and 106 years old further supports this pattern, with vascular diseases remaining the leading cause of death. These findings highlight that even exceptional longevity usually ends with a specific disease.
What Actually Causes Death in Aging Across Species
The main causes of death vary significantly between species. In laboratory mice, cancer dominates and accounts for about 84 to 89 percent of deaths associated with aging. Dogs show a similar trend, with nearly half of older dogs dying from cancer. Among captive nonhuman primates, the pattern looks closer to humans, as cardiovascular disease causes more than 60 percent of deaths in aging rhesus macaques. Even small invertebrates display species-specific limits on lifespan. Intestinal or neuromuscular failure often limits survival in Drosophila, while pharyngeal infections and deterioration drive mortality in C. elegans.
“This pattern illustrates that interventions targeting specific pathologies can extend lifespan by addressing critical bottlenecks to survival, but they do not necessarily slow the overall aging process,” the authors write.
Understanding this distinction is important when considering how human lifespan has increased over the past two centuries. In the past, infectious diseases such as the bubonic plague, smallpox, and tuberculosis were the main causes of death and killed millions of people. Advances in medicine and public health, including vaccines, antibiotics, and sanitation improvements, greatly reduced deaths from these diseases.
Lifespan Gains Do Not Always Mean Slower Aging
Yet according to the researchers, this historical shift reflects changing causes of death rather than a slowdown in the biological pace of aging. By reducing deaths from infectious disease, society delayed when people die, but did not necessarily alter the underlying processes that drive aging.
This observation has important implications for modern aging research. If lifespan increases simply because one major disease is delayed, then longer survival does not automatically mean that aging itself has slowed. For example, an intervention that extends the lifespan of mice by delaying cancer is fundamentally different from one that slows the gradual decline of many body systems, even if both produce similar survival curves.
Epigenetic aging clocks, which estimate biological age using DNA methylation patterns, have become widely used tools in aging research. They can help classify individuals by risk level, detect accelerated aging in populations, and monitor biological changes over time. However, Ehninger and Keshavarz argue that these tools raise more profound questions about what exactly they measure.
Limits of Epigenetic Aging Clocks
A major limitation is that aging clocks are based on correlations. They identify molecular patterns associated with chronological age, but those patterns may not directly cause aging. Instead, they may simply reflect biological changes that happen as a result of aging.
The authors compare this to estimating a person’s age from facial images. Wrinkles and gray hair can help predict age, but they reveal little about the biological mechanisms that drive aging. Supporting this concern, the review cites epigenome-wide Mendelian randomization studies showing that traditional aging clocks are not strongly enriched for CpG sites that play causal roles in aging.
Another issue is that most aging clocks capture a single moment in time. If an intervention appears to reduce biological age, researchers must determine whether aging has actually slowed or whether the treatment simply shifted biomarker values. Even newer tools such as DunedinPACE, which aim to estimate the pace of aging rather than static biological age, often rely on biomarkers linked to age-related traits without directly identifying underlying mechanisms.
Problems With Frailty Indices as Aging Measures
Frailty indices face similar challenges. These indices are typically based on small sets of semiquantitative traits, such as fur condition, spinal curvature, or the presence of tumors, scored using simple categorical scales. As a result, they represent only limited aspects of aging-related change.
Because these scores combine multiple traits into a single number, each component is implicitly treated as equally important biologically. Improvements in a single factor, such as reduced tumor burden, could lower the overall frailty score and give the impression that aging has broadly improved, even though the change reflects improvement in one specific disease.
The review also examines the hallmarks of aging framework, first introduced in 2013 and expanded to twelve hallmarks in 2023. These hallmarks include processes such as genomic instability, telomere shortening, epigenetic alterations, disrupted protein maintenance, and cellular senescence. The framework has strongly shaped research priorities and funding decisions. The authors ask whether the available evidence truly supports the idea that targeting these hallmarks slows aging.
Do the Hallmarks of Aging Truly Drive Aging?
To investigate this question, Keshavarz and Ehninger analyzed the primary studies cited as evidence for each hallmark. Their review focused on research used to claim causal relationships between these processes and aging.
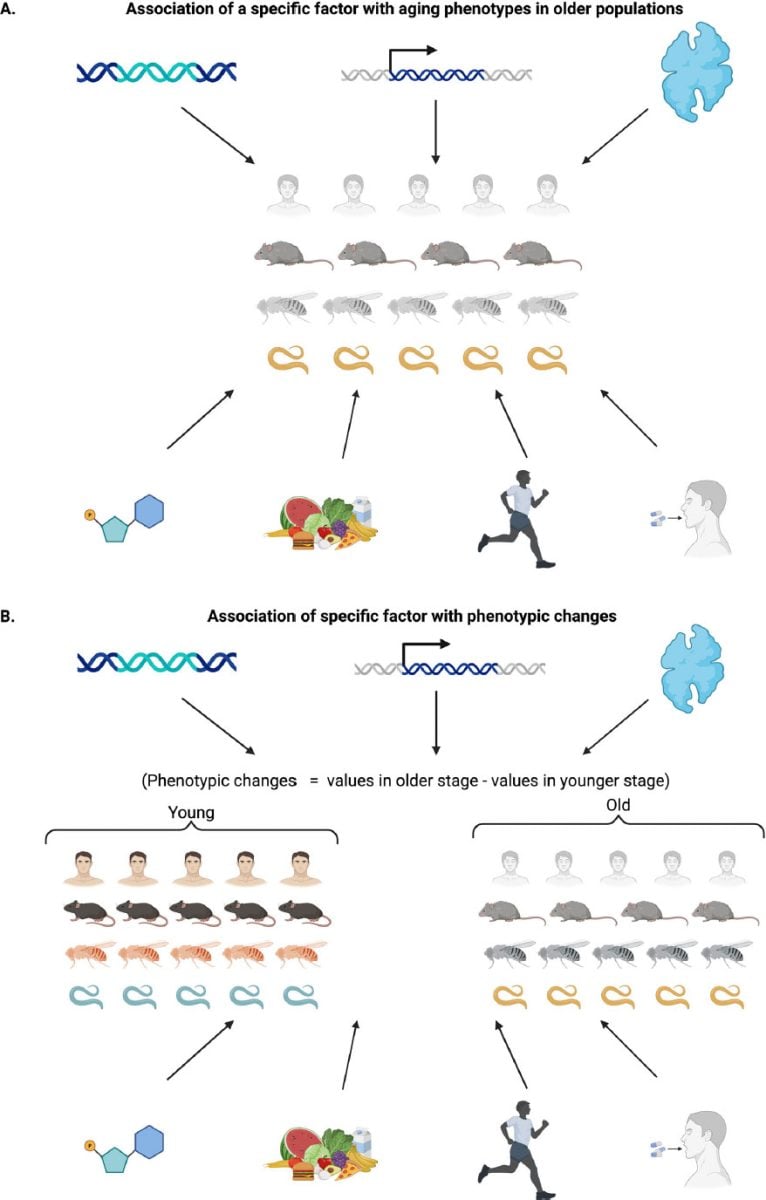
They found a major methodological limitation. Between 56.86 and 99.96 percent of the phenotypes used as supporting evidence were examined only in older animals. Researchers often did not test the same interventions in young treated groups. Without these comparisons, it is impossible to determine whether treatments actually slow aging or simply create age-independent baseline effects.
When studies did include young animals, interventions often affected both age groups. Across the research cited for the hallmarks framework, the authors identified 602 phenotypes tested in young animals. Of these, 436, or 72.4 percent, showed similar effects in young groups. This suggests that many observed changes reflect baseline physiological shifts rather than true antiaging mechanisms.
“Consequently, the evidence cited for most hallmarks supports the presence of general physiological effects rather than true antiaging mechanisms,” the review concludes.
Distinguishing True Antiaging Effects From Baseline Changes
The researchers propose a framework to better distinguish genuine aging interventions from general physiological effects. They describe three categories of treatment outcomes for age-sensitive phenotypes.
Rate effects occur when an intervention reduces the slope of age-related change, which would indicate that it is modifying the processes driving aging. Baseline effects occur when similar changes appear in both young and old animals, suggesting the treatment works independently of aging. Mixed effects occur when both groups change, but older animals show stronger responses, which requires careful interpretation because it may reflect combined mechanisms or differences in treatment duration.
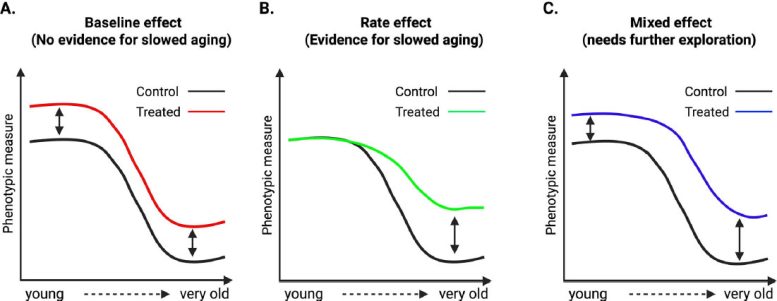
To illustrate these categories, the review highlights studies of well-known lifespan extending interventions. These include intermittent fasting, rapamycin, and genetic changes affecting mTOR and growth hormone signaling. Researchers performed deep phenotyping on both young and old animals receiving these treatments.
Although these interventions are known to extend lifespan, many of the measured traits changed similarly in both age groups. This indicates baseline shifts rather than slower age-related decline. In other words, the treatments altered phenotype values but did not consistently change the rate of aging.
Major Unanswered Questions About Aging Biology
The analysis also raises broader questions about how aging works across the body. For instance, scientists still do not fully understand why some tissues age faster than others or how strongly aging processes are coordinated between organs.
Evidence shows that different tissues follow distinct aging trajectories. These differences may arise from developmental patterns and lifelong variation in cell turnover, metabolic activity, and exposure to environmental stress. Researchers also continue to debate whether aging is controlled mainly by central regulatory systems or by processes that occur independently within individual cells.
Another challenge involves translating findings between species. The dominant causes of death vary widely across organisms. Humans mainly die from cardiovascular disease, mice from cancer, fish from infections, flies from intestinal or neuromuscular failure, and worms from bacterial infection. These differences suggest that aging may not operate as a single universal process but instead as a complex mix of species-specific and tissue-specific mechanisms shaped by evolution and environment.
The consequences of these findings extend beyond academic debates. If commonly used aging markers confuse baseline physiological effects with real changes in aging, research resources could be directed toward treatments that improve symptoms without altering the underlying biology of aging. The authors emphasize that geroscience aims to understand mechanisms that drive age-related changes in physiology. Other scientific fields already focus on regulating specific biological functions such as hormones, neural activity, or immune responses.
Implications for Geroscience and Drug Development
For example, a therapy that improves cognitive performance at any age may still be valuable. However, it cannot be considered a treatment for cognitive aging unless it clearly slows the rate of cognitive decline over time. This distinction has major implications for drug development, clinical trial design, and expectations for patients seeking therapies that truly modify aging.
The researchers conclude with several recommendations for improving study design. Scientists should develop standardized panels of age-sensitive phenotypes across multiple tissues and organ systems, covering molecular, cellular, tissue, and whole-organism levels. Experiments should include both young-treated and old-treated groups so that researchers can identify whether interventions change the rate of aging or simply shift baseline biology.
They also recommend classifying outcomes into rate effects, baseline effects, or mixed effects rather than assuming all observed changes reflect aging modification. Researchers should map age-sensitive trajectories to select appropriate time points for measurement while avoiding survival bias. Finally, claims about systemic antiaging effects should be supported by evidence across many phenotypes and tissues rather than based on improvements in a single outcome.
“Refining both discovery pipelines and intervention testing frameworks will support a more mechanistic understanding of aging by enabling researchers to distinguish between interventions that simply extend lifespan or improve isolated age-sensitive phenotypes and those that fundamentally modify the biological processes driving age-related decline,” the authors write.
Reference: “Beyond the hallmarks of aging: Rethinking what aging is and how we measure it” by Maryam Keshavarz and Dan Ehninger, 2 December 2025, Genomic Psychiatry.
DOI: 10.61373/gp025i.0119
The work was supported by the ETERNITY project consortium, funded by the European Union through the Horizon Europe Marie Sklodowska-Curie Actions Doctoral Networks under grant agreement number 101072759.
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.
Source link

