Scientists Solve a Mystery Behind Your Sense of Touch

A new study from Scripps Research reveals how a key touch-sensing protein detects mechanical forces with remarkable precision.
Each time something lightly presses against your skin, specialized sensory neurons translate that mechanical force into electrical signals that the brain interprets as touch. Researchers have long known that a protein called PIEZO2 plays a central role in detecting touch. What remained unclear, however, was why PIEZO2 responds best to small, localized forces experienced by sensory neurons, while its closely related counterpart, PIEZO1, reacts to broader mechanical stress such as the stretching of cells in blood vessels.
A new study from Scripps Research now provides answers. The research, published in Nature, explains how PIEZO2 recognizes specific mechanical forces and why it may have evolved as the body’s primary detector of gentle touch. The findings could also help scientists better understand sensory disorders linked to mutations in the PIEZO2 gene.
“Touch is one of our most fundamental senses, yet we didn’t fully understand how it’s processed at the molecular level,” says co-senior author Professor Ardem Patapoutian, the Presidential Endowed Chair in Neurobiology at Scripps Research and a Howard Hughes Medical Institute Investigator. “We wanted to see how the structure of PIEZO2 shapes what a cell can actually feel.”
A Nobel Prize–Winning Discovery
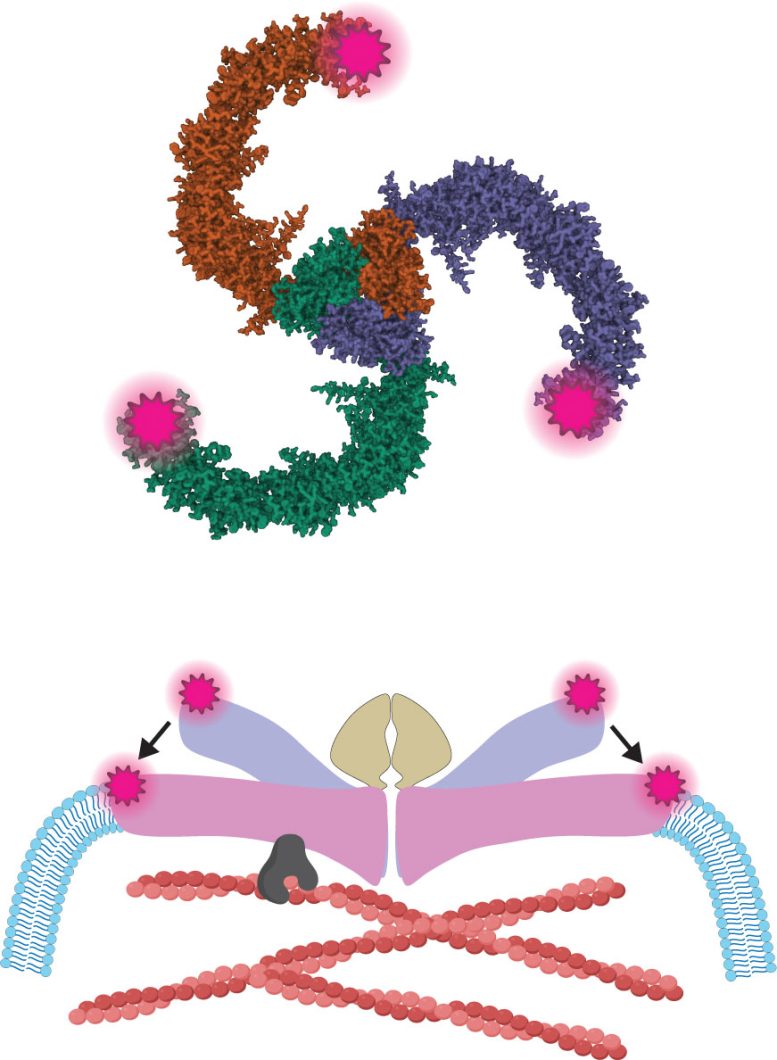
Patapoutian shared the 2021 Nobel Prize in Physiology or Medicine for discovering PIEZO1 and PIEZO2. These proteins function as ion channels, which are microscopic gateways embedded in the membranes of cells. They open when mechanical force is applied.
Once these channels open, charged particles move into the cell. This movement generates electrical signals that allow the nervous system to perceive sensations such as touch, body position, and certain forms of pain.
Although molecular models show PIEZO1 and PIEZO2 as nearly identical in structure, their behavior in living cells is very different. PIEZO2 is particularly important in the somatosensory nervous system, the network of neurons responsible for detecting touch. These neurons are extremely sensitive to tiny indentations, such as a gentle tap on the skin.
PIEZO1, in contrast, responds more strongly when a cell membrane is stretched. This type of force can occur when a cell swells or is pulled, rather than when it is pressed at a single point.
Imaging Proteins Moving Inside Living Cells
To understand why these two channels respond differently, the researchers turned to minimal fluorescence photon flux (MINFLUX) super-resolution microscopy. Imaging support came from Professor Scott Henderson, who directs the Scripps Research Core Microscopy Facility, along with Senior Staff Scientist Kathryn Spencer.
Other methods, including cryogenic electron microscopy (cryo-EM), can produce detailed structural images of frozen PIEZO proteins that reveal their overall shape. However, these images represent static snapshots. MINFLUX allows researchers to monitor the location and movement of proteins inside living cells with nanometer-scale precision. A nanometer is one billionth of a meter (about 3.9 × 10⁻⁸ inches), roughly 100,000 times smaller than the width of a human hair.
“Cryo-EM gives us beautiful structural snapshots, but it can’t show us how a protein moves in its native cellular environment,” notes first and co-senior author Eric Mulhall, a postdoctoral fellow in Patapoutian’s lab.
“What I love about this work, led by Eric Mulhall, is that it connects discoveries across an unusually wide range of scales,” adds Patapoutian. “It’s one of the few studies I’ve seen that spans from nanometer-scale super-resolution microscopy all the way to ex vivo and in vivo experiments, linking single-molecule insights to physiological function.”
A Physical Link to the Cell’s Internal Scaffold
Using MINFLUX along with electrical recordings that measure ion flow, the team observed how PIEZO2 changed shape when force was applied. Those electrical recordings, carried out by second author and Staff Scientist Oleg Yarishkin, allowed a direct connection between PIEZO2’s structural changes and channel activity. The team found that PIEZO2 was intrinsically stiffer than PIEZO1 and physically connected (or “tethered”) to the cell’s internal scaffolding, known as the actin cytoskeleton. The cytoskeleton is a network of protein fibers called actin filaments that helps maintain cell shape and transmit forces.
Tethering occurs through a protein called filamin-B, which connects membrane proteins to actin filaments. When a cell was poked, this internal link helped convey force to PIEZO2, making the channel more likely to open. However, simple membrane stretching didn’t activate PIEZO2 when the tether was intact.
The team identified the specific region where PIEZO2 connected to filamin-B and showed that disrupting this connection changed how the channel sensed force. In mouse sensory neurons—the nerve cells responsible for detecting touch—removing the tether reduced PIEZO2’s sensitivity to indentation and unexpectedly allowed the channel to respond to membrane stretch, a type of force it would normally ignore.
“We were surprised by how differently the two channels responded to the same type of force,” recalls Mulhall. “Membrane stretch expands and activates PIEZO1, though we observed the opposite response in PIEZO2. This was a strong indication that these channels operate through distinct mechanisms.”
Implications for Sensory Disorders
The findings suggest that cells can fine-tune their sensitivity to touch not only by choosing which ion channel to use, but also by controlling how that channel is physically integrated within a cell. Because filamin-B is widely expressed across tissues, tethering may help tailor PIEZO2 for registering gentle, everyday touch. Understanding this mechanism could also shed light on what happens when it’s impaired.
Mutations in PIEZO2 can cause sensory disorders affecting touch and body awareness, while mutations in filamin-B are associated with skeletal and developmental conditions. By clarifying how these proteins interact, the study provides a clearer framework for interpreting such genetic findings and guiding future research into sensory function.
“Our results shift the perspective on how touch begins at the molecular level,” explains Patapoutian. “A protein’s physical connections inside a cell determine what kinds of forces it can sense. That’s a new way of thinking about how we feel the world around us.”
Reference: “The molecular basis of force selectivity by PIEZO2” by Eric M. Mulhall, Oleg Yarishkin, Rose Z. Hill, Anna K. Koster and Ardem Patapoutian, 4 March 2026, Nature.
DOI: 10.1038/s41586-026-10182-7
This work was supported by funding from the National Institutes of Health (grants K99 GM155547 and R35 NS105067), a postdoctoral fellowship from the George E. Hewitt Foundation for Medical Research, the Howard Hughes Medical Institute Investigator Program, and The Warren Alpert Distinguished Scholar Award in Neuroscience.
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.
Source link

