Scientists Uncover the Immune System’s Hidden Role
Why do diabetic wounds refuse to heal? A new scientific review highlights how disruptions in the timing and behavior of immune cells may hold the key to understanding this widespread medical challenge.
Chronic diabetic ulcers are among the most serious and costly complications associated with diabetes. More than 131 million people around the world are affected, and treatment costs reach an estimated $755 billion each year. These wounds often lead to amputation and carry a high risk of death, highlighting the urgent need to better understand the biological processes that prevent them from healing.
The immune system plays a central role in coordinating wound repair. However, many studies have not fully examined how different immune cell populations change over time in diabetic wounds. A comprehensive review led by Yi Ru and colleagues addresses this gap. The researchers systematically analyzed how a wide range of immune cells behave during the different stages of diabetic wound healing.
The review examines immune cells involved in wound healing and how diabetes disrupts their functions, with particular focus on monocytes and macrophages.
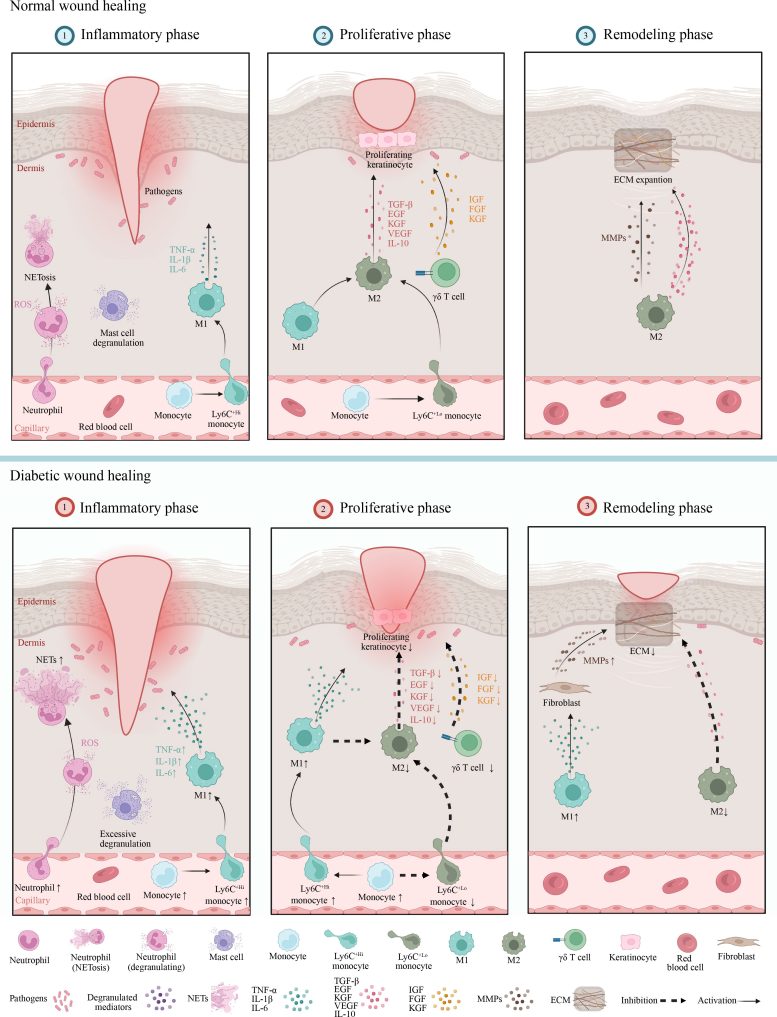
Normally, monocytes become macrophages that shift from a pro-inflammatory M1 state to a pro-repair M2 state, helping resolve inflammation and promote tissue repair.
In diabetic wounds, this transition does not occur properly. Macrophages often remain stuck in a pro-inflammatory state, which prolongs inflammation and promotes tissue damage rather than healing.
Neutrophils and Mast Cells in Chronic Inflammation
Neutrophils are typically the first immune cells to reach an injury. They play a key role in destroying invading microbes and triggering the early inflammatory response. In diabetic wounds, however, neutrophil extracellular trap formation becomes abnormal.
This dysregulation leads to prolonged inflammation and tissue injury. Neutrophils release excessive amounts of enzymes and reactive oxygen species, which can damage surrounding tissue and slow the healing process.
Mast cells also contribute to wound repair. These cells release substances such as histamine, serotonin, and various growth factors that influence blood vessel permeability and attract additional immune cells to the wound.
According to the review, mast cell activity differs greatly between normal and diabetic wounds. In diabetes, mast cells can become excessively activated, which may help sustain the long-lasting inflammatory environment seen in chronic ulcers.
Dendritic cells play a critical role in connecting the innate and adaptive branches of the immune system. In the skin, this group includes Langerhans cells in the epidermis and dendritic cells located within the dermis.
Recent studies show that dendritic cells in diabetic wounds struggle to perform efferocytosis, the process of clearing dying cells. When this cleanup process fails, dead cells and debris accumulate in the wound.
The buildup of cellular debris continues to trigger inflammatory signals and interferes with healing. Researchers have identified the SLC7A11 transporter as an important regulator of this process. In people with diabetes, reduced activity of this transporter appears to impair dendritic cell function and hinder wound recovery.
The Role of T Cells in Repair
T lymphocytes also participate in regulating inflammation and supporting tissue repair. Among these are regulatory T cells and γδ T cells, both of which influence the healing environment.
Regulatory T cells help control inflammation and encourage the production of factors that support tissue rebuilding. In diabetic wounds, these cells are fewer in number and often function less effectively, which contributes to slower healing.
Another important group is dendritic epidermal T cells, a specialized γδ T cell population found in the skin. These cells produce insulin like growth factor 1 and other molecules that stimulate keratinocyte growth and help close wounds.
In diabetic conditions, dendritic epidermal T cells show reduced activation and produce fewer signaling molecules, further weakening the healing response.
B cells and natural killer cells are less studied in the context of wound healing, but they also appear to play meaningful roles. B cells influence repair by producing antibodies and by affecting how macrophages shift between inflammatory and reparative states. Recent findings suggest that B cell recruitment may encourage M2 macrophage polarization, which helps limit excessive inflammation during the healing phase.
Natural killer cells regulate inflammation and blood vessel formation through cytokine signaling and interactions with other immune cells. Scientists are still working to clarify their exact contributions to diabetic wound repair.
Immune Based Treatment Strategies
Understanding how immune cells behave in diabetic wounds has important implications for treatment. The review highlights several emerging approaches designed to regulate immune activity and improve healing.
One strategy involves topical anti-cytokine biologic treatments that interrupt persistent inflammatory signaling in chronic wounds. Therapies that influence macrophage polarization are particularly promising. Several drugs have shown the ability to promote the M2 macrophage state and speed up wound closure.
Mesenchymal stem cell therapies and the extracellular vesicles they release can provide multiple immune-regulating effects. At the same time, new biomaterials and advanced wound dressings are being designed to deliver immune-regulating compounds directly to the wound site.
Emerging Therapeutic Strategies
Researchers are also exploring several new technologies. These include Janus liposozyme systems that help regulate redox balance and immune responses, drugs that control macrophage behavior and have shown success in randomized clinical trials, and IL 15 superagonists that strengthen the activity of dendritic epidermal T cells.
The review emphasizes that timing is critical in diabetic wound treatment. Different immune environments exist during the inflammatory, proliferative, and remodeling stages of healing, so therapies must be carefully matched to the appropriate stage.
Looking ahead, the authors highlight the potential for personalized treatment strategies based on immune profiling of individual wounds. Combination therapies that target multiple immune cell populations at the same time may also prove effective.
The review also calls for further research to clarify ongoing debates about macrophage classification systems. A deeper understanding of how immune cells interact and change throughout the healing process may ultimately lead to more precise and effective treatments for this serious complication of diabetes.
Reference: “Immune cells in diabetic wound repair: the key to better wound management” by Yi Ru, Yunxi Cai, Guangyuan Cheng, Xiaoxuan Ma, Jingsi Jiang, Jiankun Song, Ying Luo, Ying Zhang, Qi Zheng, Mingxia Wang, Chunjie Gao, Bin Li, Le Kuai, Yue Luo and Zhan Zhang, 27 December 2025, MedScience.
DOI: 10.1007/s11684-025-1190-y
Never miss a breakthrough: Join the SciTechDaily newsletter.
Follow us on Google and Google News.
Source link

