Kwok KHM, Lam KSL, Xu A. Heterogeneity of white adipose tissue: molecular basis and clinical implications. Exp Mol Med. 2016;48:e215.
Lowell BB, Spiegelman BM. Towards a molecular understanding of adaptive thermogenesis. Nature. 2000;404:652–60.
Scheja L, Heeren J. The endocrine function of adipose tissues in health and cardiometabolic disease. Nat Rev Endocrinol. 2019;15:507–24.
Bartelt A, Heeren J. Adipose tissue browning and metabolic health. Nat Rev Endocrinol. 2014;10:24–36.
Harms M, Seale P. Brown and beige fat: development, function and therapeutic potential. Nat Med. 2013;19:1252–63.
Schulz TJ, Huang P, Huang TL, Xue R, McDougall LE, Townsend KL, et al. Brown-fat paucity due to impaired BMP signalling induces compensatory browning of white fat. Nature. 2013;495:379–83.
Cohen P, Kajimura S. The cellular and functional complexity of thermogenic fat. Nat Rev Mol Cell Biol. 2021;22:393–409.
Brestoff JR, Artis D. Immune regulation of metabolic homeostasis in health and disease. Cell. 2015;161:146–60.
Wu J, Cohen P, Spiegelman BM. Adaptive thermogenesis in adipocytes: is beige the new brown?. Genes Dev. 2013;27:234–50.
Scheja L, Heeren J. Metabolic interplay between white, beige, brown adipocytes and the liver. J Hepatol. 2016;64:1176–86.
Wang B, Hu Z, Cui L, Zhao M, Su Z, Jiang Y, et al. βAR-mTOR-lipin1 pathway mediates PKA-RIIβ deficiency-induced adipose browning. Theranostics. 2024;14:5316–35.
Wang B, Zhao M, Su Z, Jin B, Yang X, Zhang C, et al. RIIβ-PKA in GABAergic neurons of dorsal median hypothalamus governs white adipose browning. Adv Sci. 2022;10:2205173.
Guo B, Liu J, Wang B, Zhang C, Su Z, Zhao M, et al. Withaferin a promotes white adipose browning and prevents obesity through sympathetic nerve–activated Prdm16-FATP1 axis. Diabetes. 2021;71:249–63.
Wang Q, Li H, Tajima K, Verkerke ARP, Taxin ZH, Hou Z, et al. Post-translational control of beige fat biogenesis by PRDM16 stabilization. Nature. 2022;609:151–8.
Boström P, Wu J, Jedrychowski MP, Korde A, Ye L, Lo JC, et al. A PGC1-α-dependent myokine that drives brown-fat-like development of white fat and thermogenesis. Nature. 2012;481:463–8.
Wang T, Sharma AK, Wu C, Maushart CI, Ghosh A, Yang W, et al. Single-nucleus transcriptomics identifies separate classes of UCP1 and futile cycle adipocytes. Cell Metab. 2024;36:2130–45.e7.
Czernichow S, Ziegler O, Clément K, Coupaye M, Laville M. Assessing worldwide trends of underweight and obesity. Lancet. 2024;404:1642–3.
Polyzos SA, Kountouras J, Mantzoros CS. Obesity and nonalcoholic fatty liver disease: From pathophysiology to therapeutics. Metabolism. 2019;92:82–97.
Pasarica M, Sereda OR, Redman LM, Albarado DC, Hymel DT, Roan LE, et al. Reduced adipose tissue oxygenation in human obesity: evidence for rarefaction, macrophage chemotaxis, and inflammation without an angiogenic response. Diabetes. 2009;58:718–25.
Hagberg CE, Spalding KL. White adipocyte dysfunction and obesity-associated pathologies in humans. Nat Rev Mol Cell Biol. 2024;25:270–89.
Becher T, Palanisamy S, Kramer DJ, Eljalby M, Marx SJ, Wibmer AG, et al. Brown adipose tissue is associated with cardiometabolic health. Nat Med. 2021;27:58–65.
Swinburn BA, Sacks G, Hall KD, McPherson K, Finegood DT, Moodie ML, et al. The global obesity pandemic: shaped by global drivers and local environments. Lancet. 2011;378:804–14.
Haase CL, Eriksen KT, Lopes S, Satylganova A, Schnecke V, McEwan P. Body mass index and risk of obesity-related conditions in a cohort of 2.9 million people: evidence from a UK primary care database. Obes Sci Pr. 2021;7:137–47.
O’Mara AE, Johnson JW, Linderman JD, Brychta RJ, McGehee S, Fletcher LA, et al. Chronic mirabegron treatment increases human brown fat, HDL cholesterol, and insulin sensitivity. J Clin Invest. 2020;130:2209–19.
Li HF, He TL, Ma DJ, Gong HH, Yang ZG. Circular RNAs and mammalian follicular development: current insights and future prospects—an updated review. Cell Death Discov. 2025;11:454–67.
He TL, Chen QY, Tian K, Xia YZ, Dong GZ, Yang ZG. Functional role of circRNAs in the regulation of fetal development, muscle development, and lactation in livestock. BioMed Res Int. 2021;2021:5383210.
Salzman J, Gawad C, Wang PL, Lacayo N, Brown PO. Circular RNAs are the predominant transcript isoform from hundreds of human genes in diverse cell types. PloS One. 2012;7:e30733.
Kristensen LS, Andersen MS, Stagsted LVW, Ebbesen KK, Hansen TB, Kjems J. The biogenesis, biology and characterization of circular RNAs. Nat Rev Genet. 2019;20:675–91.
Ashwal-Fluss R, Meyer M, Pamudurti NR, Ivanov A, Bartok O, Hanan M, et al. circRNA biogenesis competes with pre-mRNA splicing. Mol Cell. 2014;56:55–66.
Yang L, Wilusz JE, Chen LL. Biogenesis and regulatory roles of circular RNAs. Annu Rev Cell Dev Biol. 2022;38:263–89.
Chen LL. The expanding regulatory mechanisms and cellular functions of circular RNAs. Nat Rev Mol Cell Biol. 2020;21:475–90.
Memczak S, Jens M, Elefsinioti A, Torti F, Krueger J, Rybak A, et al. Circular RNAs are a large class of animal RNAs with regulatory potency. Nature. 2013;495:333–8.
Hansen TB, Jensen TI, Clausen BH, Bramsen JB, Finsen B, Damgaard CK, et al. Natural RNA circles function as efficient microRNA sponges. NATURE. 2013;495:384–8.
Harland R, Misher L. Stability of RNA in developing Xenopus embryos and identification of a destabilizing sequence in TFIIIA messenger RNA. Dev Camb Engl. 1988;102:837–52.
Yang ZG, He TL, Chen QY. The roles of CircRNAs in regulating muscle development of livestock animals. Front Cell Dev Biol. 2021;9:619329.
Zhang X, Chen L, Xiao B, Liu H, Su Y. Circ_0075932 in adipocyte-derived exosomes induces inflammation and apoptosis in human dermal keratinocytes by directly binding with PUM2 and promoting PUM2-mediated activation of AuroraA/NF-κB pathway. Biochem Biophys Res Commun. 2019;511:551–8.
He TL, Yuan ZD, Chen QY, Luo J, Mao JN, Yang YL, et al. Circular RNAs mediate the regulation of maternal placental nutrient transport and fetal development by sugar-sweetened beverages. Food Res Int. 2024;193:114856.
He TL, Chen QY, Yuan ZD, Yang YL, Cao K, Luo J, et al. Effects of maternal high-fat diet on fetal growth, placental nutrient transporters and circular RNA expression profiles. Food Funct. 2023;14:9391–406.
He TL, Yuan ZD, Chen QY, Luo J, Mao JN, Tang ZR, et al. Circular RNAs mediate the effects of dietary tryptophan on the transformation of muscle fiber types in weaned piglets. J Agric Food Chem. 2024;72:8595–605.
Zhou WY, Cai ZR, Liu J, Wang DS, Ju HQ, Xu RH. Circular RNA: metabolism, functions and interactions with proteins. Mol Cancer. 2020;19:172.
Liu B, Yang G, Wang X, Liu J, Lu Z, Wang Q, et al. CircBACH1 (hsa_circ_0061395) promotes hepatocellular carcinoma growth by regulating p27 repression via HuR. J Cell Physiol. 2020;235:6929–41.
Du WW, Yang W, Liu E, Yang Z, Dhaliwal P, Yang BB. Foxo3 circular RNA retards cell cycle progression via forming ternary complexes with p21 and CDK2. Nucleic Acids Res. 2016;44:2846–58.
Li Q, Wang Y, Wu S, Zhou Z, Ding X, Shi R, et al. CircACC1 regulates assembly and activation of AMPK complex under metabolic stress. Cell Metab. 2019;30:157–173.e7.
Placing non-coding RNAs in the Central Dogma: An interview with Ling-Ling Chen. Mol Cell. 2023;83:317–9.
Li Z, Huang C, Bao C, Chen L, Lin M, Wang X, et al. Exon-intron circular RNAs regulate transcription in the nucleus. Nat Struct Mol Biol. 2017;24:194–194.
Yang Y, Wang Z. IRES-mediated cap-independent translation, a path leading to hidden proteome. J Mol Cell Biol. 2019;11:911–9.
Yang Y, Fan X, Mao M, Song X, Wu P, Zhang Y, et al. Extensive translation of circular RNAs driven by N6-methyladenosine. Cell Res. 2017;27:626–41.
Wesselhoeft RA, Kowalski PS, Anderson DG. Engineering circular RNA for potent and stable translation in eukaryotic cells. Nat Commun. 2018;9:2629.
Abe N, Matsumoto K, Nishihara M, Nakano Y, Shibata A, Maruyama H, et al. Rolling circle translation of circular RNA in living human cells. Sci Rep. 2015;5:16435.
Huang W, Ling Y, Zhang S, Xia Q, Cao R, Fan X, et al. TransCirc: an interactive database for translatable circular RNAs based on multi-omics evidence. Nucleic Acids Res. 2021;49:D236–42.
Sun L, Wang W, Han C, Huang W, Sun Y, Fang K, et al. The oncomicropeptide APPLE promotes hematopoietic malignancy by enhancing translation initiation. Mol Cell. 2021;81:4493–4508.e9.
Wen S, yang, Qadir J, Yang BB. Circular RNA translation: novel protein isoforms and clinical significance. TRENDS Mol Med. 2022;28:405–20.
Ho-Xuan H, Glažar P, Latini C, Heizler K, Haase J, Hett R, et al. Comprehensive analysis of translation from overexpressed circular RNAs reveals pervasive translation from linear transcripts. Nucleic Acids Res. 2020;48:10368–82.
Huang W, Chen TQ, Fang K, Zeng ZC, Ye H, Chen YQ. N6-methyladenosine methyltransferases: functions, regulation, and clinical potential. J Hematol OncolJ Hematol Oncol. 2021;14:117.
Yang Y, Gao X, Zhang M, Yan S, Sun C, Xiao F, et al. Novel role of FBXW7 circular RNA in repressing glioma tumorigenesis. J Natl Cancer Inst. 2018;110:304–15.
Wilusz JE, Sharp PA. Molecular biology. A circuitous route to noncoding RNA. Science. 2013;340:440–1.
Liu K, Liu X, Deng Y, Li Z, Tang A. CircRNA-mediated regulation of brown adipose tissue adipogenesis. Front Nutr. 2022;9:926024.
Huang Z, Mou T, Luo Y, Pu X, Pu J, Wan L, et al. Inhibition of miR-450b-5p ameliorates hepatic ischemia/reperfusion injury via targeting CRYAB. Cell Death Dis. 2020;11:455.
Huang Z, Zheng D, Pu J, Dai J, Zhang Y, Zhang W, et al. MicroRNA-125b protects liver from ischemia/reperfusion injury via inhibiting TRAF6 and NF-κB pathway. Biosci Biotechnol Biochem. 2019;83:829–35.
Huang W, Jiang L, Jiang Y, Li S, Liu W, Zong K, et al. ANGPTL4 induces Kupffer cell M2 polarization to mitigate acute rejection in liver transplantation. Sci Rep. 2025;15:986.
Shao J, Wang M, Zhang A, Liu Z, Jiang G, Tang T, et al. Interference of a mammalian circRNA regulates lipid metabolism reprogramming by targeting miR-24-3p/Igf2/PI3K-AKT-mTOR and Igf2bp2/Ucp1 axis. Cell Mol LIFE Sci. 2023;80:252.
Shen X, Tang J, Huang Y, Lan X, Lei C, Chen H. CircRNF111 Contributes to Adipocyte Differentiation by Elevating PPARγ Expression via miR-27a-3p. Epigenetics. 2023;18:2145058.
Wu J, Zhang S, Yue B, Zhang S, Jiang E, Chen H, et al. CircRNA profiling reveals CircPPARγ modulates adipogenic differentiation via sponging miR-92a-3p. J Agric Food Chem. 2022;70:6698–708.
Zhang S, Jiang E, Kang Z, Bi Y, Liu H, Xu H, et al. CircRNA profiling reveals an abundant circBDP1 that regulates bovine fat development by sponging miR-181b/miR-204 targeting Sirt1/TRARG1. J Agric Food Chem. 2022;70:14312–28.
Tian W, Liu Y, Zhang W, Nie R, Ling Y, Zhang B, et al. CircDOCK7 facilitates the proliferation and adipogenic differentiation of chicken abdominal preadipocytes through the gga-miR-301b-3p/ACSL1 axis. J Anim Sci Biotechnol. 2023;14:91.
Song XH, He N, Xing YT, Jin XQ, Li YW, Liu SS, et al. A novel age-related circular RNA Circ-ATXN2 inhibits proliferation, promotes cell death and adipogenesis in rat adipose tissue-derived stromal cells. Front Genet. 2021;12:761926.
Liu Y, Liu H, Li Y, Mao R, Yang H, Zhang Y, et al. Circular RNA SAMD4A controls adipogenesis in obesity through the miR-138-5p/EZH2 axis. Theranostics. 2020;10:4705–19.
Zhang T, Zhang Z, Xia T, Liu C, Sun C. circNrxn2 Promoted WAT Browning via Sponging miR-103 to Relieve Its Inhibition of FGF10 in HFD Mice. Mol Ther Nucleic Acids. 2019;17:551–62.
Zhong J, Xu A, Xu P, Su M, Wang P, Liu Z, et al. Circ_0000235 targets MCT4 to promote glycolysis and progression of bladder cancer by sponging miR-330-5p. Cell Death Discov. 2023;9:1–16.
Zhu Y, Gui W, Lin X, Li H. Knock-down of circular RNA H19 induces human adipose-derived stem cells adipogenic differentiation via a mechanism involving the polypyrimidine tract-binding protein 1. Exp Cell Res. 2020;387:111753.
Yu X, Tong H, Chen J, Tang C, Wang S, Si Y, et al. CircRNA MBOAT2 promotes intrahepatic cholangiocarcinoma progression and lipid metabolism reprogramming by stabilizing PTBP1 to facilitate FASN mRNA cytoplasmic export. Cell Death Dis. 2023;14:20.
Xia H, Wu Y, Zhao J, Cheng C, Lin J, Yang Y, et al. N6-Methyladenosine-modified circSAV1 triggers ferroptosis in COPD through recruiting YTHDF1 to facilitate the translation of IREB2. Cell Death Differ. 2023;30:1293–304.
Li J, Wang X, Shi L, Liu B, Sheng Z, Chang S, et al. A mammalian conserved circular RNA CircLARP1B regulates hepatocellular carcinoma metastasis and lipid metabolism. Adv Sci Weinh Baden-Wurtt Ger. 2024;11:e2305902.
Zhu T, Cen Y, Chen Z, Zhang Y, Zhao L, Wang J, et al. Oncogenic circTICRR suppresses autophagy via binding to HuR protein and stabilizing GLUD1 mRNA in cervical cancer. Cell Death Dis. 2022;13:1–13.
Wang Q, Kajimura S. Naa10P puts a brake on PGC1α and fat browning. Nat Struct Mol Biol. 2019;26:849–51.
Huang A, Zheng H, Wu Z, Chen M, Huang Y. Circular RNA-protein interactions: functions, mechanisms, and identification. Theranostics. 2020;10:3503–17.
Zhu R, Feng Y, Yang X, Li R, Song Z, Liu Q, et al. Functionally conserved PPARG exonic circRNAs enhance intramuscular fat deposition by regulating PPARG and HSL. Int J Biol Macromol. 2024;257:128613.
Wu L, Zhou L, Chen C, Gong J, Xu L, Ye J, et al. Cidea controls lipid droplet fusion and lipid storage in brown and white adipose tissue. Sci China Life Sci. 2014;57:107–16.
Arcinas C, Tan W, Fang W, Desai TP, Teh DCS, Degirmenci U, et al. Adipose circular RNAs exhibit dynamic regulation in obesity and functional role in adipogenesis. Nat Metab. 2019;1:688–703.
Yang Y, Luo D, Shao Y, Shan Z, Liu Q, Weng J, et al. circCAPRIN1 interacts with STAT2 to promote tumor progression and lipid synthesis via upregulating ACC1 expression in colorectal cancer. Cancer Commun Lond Engl. 2023;43:100–22.
Chen N, Zhao G, Yan X, Lv Z, Yin H, Zhang S, et al. A novel FLI1 exonic circular RNA promotes metastasis in breast cancer by coordinately regulating TET1 and DNMT1. Genome Biol. 2018;19:218.
Yang ZG, Awan FM, Du WW, Zeng Y, Lyu J, Wu D, et al. The Circular RNA Interacts with STAT3, Increasing Its Nuclear Translocation and Wound Repair by Modulating Dnmt3a and miR-17 Function. Mol Ther. 2017;25:2062–74.
Jie M, Wu Y, Gao M, Li X, Liu C, Ouyang Q, et al. CircMRPS35 suppresses gastric cancer progression via recruiting KAT7 to govern histone modification. Mol Cancer. 2020;19:56.
Santoni G, Astori S, Leleu M, Glauser L, Zamora SA, Schioppa M, et al. Chromatin plasticity predetermines neuronal eligibility for memory trace formation. Science. 2024;385:eadg9982.
Chen LL. The biogenesis and emerging roles of circular RNAs. Nat Rev Mol Cell Biol. 2016;17:205–11.
Conn SJ, Pillman KA, Toubia J, Conn VM, Salmanidis M, Phillips CA, et al. The RNA binding protein quaking regulates formation of circRNAs. Cell. 2015;160:1125–34.
Jia R, Xiao MS, Li Z, Shan G, Huang C. Defining an evolutionarily conserved role of GW182 in circular RNA degradation. Cell Discov. 2019;5:45.
Dong W, Dai ZH, Liu FC, Guo XG, Ge CM, Ding J, et al. The RNA-binding protein RBM3 promotes cell proliferation in hepatocellular carcinoma by regulating circular RNA SCD-circRNA 2 production. EBioMedicine. 2019;45:155–67.
Liu CX, Chen LL. Circular RNAs: characterization, cellular roles, and applications. Cell. 2022;185:2016–34.
Meng X, Chen Q, Zhang P, Chen M. CircPro: an integrated tool for the identification of circRNAs with protein-coding potential. Bioinforma Oxf Engl. 2017;33:3314–6.
Jiang T, Xia Y, Lv J, Li B, Li Y, Wang S, et al. A novel protein encoded by circMAPK1 inhibits progression of gastric cancer by suppressing activation of MAPK signaling. Mol Cancer. 2021;20:66.
Wang Y, Tian X, Wang Z, Liu D, Zhao X, Sun X, et al. A novel peptide encoded by circ-SLC9A6 promotes lipid dyshomeostasis through the regulation of H4K16ac-mediated CD36 transcription in NAFLD. Clin Transl Med. 2024;14:e1801.
Yang F, Hu A, Guo Y, Wang J, Li D, Wang X, et al. p113 isoform encoded by CUX1 circular RNA drives tumor progression via facilitating ZRF1/BRD4 transactivation. Mol Cancer. 2021;20:123.
Qu L, Yi Z, Shen Y, Lin L, Chen F, Xu Y, et al. Circular RNA vaccines against SARS-CoV-2 and emerging variants. Cell. 2022;185:1728–44.e16.
Chen R, Wang SK, Belk JA, Amaya L, Li Z, Cardenas A, et al. Engineering circular RNA for enhanced protein production. Nat Biotechnol. 2023;41:262–72.
Jeck WR, Sorrentino JA, Wang K, Slevin MK, Burd CE, Liu J, et al. Circular RNAs are abundant, conserved, and associated with ALU repeats. RNA N Y N. 2013;19:141–57.
Zhang XO, Wang HB, Zhang Y, Lu X, Chen LL, Yang L. Complementary sequence-mediated exon circularization. Cell. 2014;159:134–47.
Ivanov A, Memczak S, Wyler E, Torti F, Porath HT, Orejuela MR, et al. Analysis of intron sequences reveals hallmarks of circular RNA biogenesis in animals. Cell Rep. 2015;10:170–7.
He Z, Ji H, Xia B, Cao X, Huang Y, Zhu Q. Invention of circRNA promoting RNA to specifically promote circRNA production. Nucleic Acids Res. 2024;52:e83.
Shi L, Yan P, Liang Y, Sun Y, Shen J, Zhou S, et al. Circular RNA expression is suppressed by androgen receptor (AR)-regulated adenosine deaminase that acts on RNA (ADAR1) in human hepatocellular carcinoma. Cell Death Dis. 2017;8:e3171.
Rybak-Wolf A, Stottmeister C, Glažar P, Jens M, Pino N, Giusti S, et al. Circular RNAs in the mammalian brain are highly abundant, conserved, and dynamically expressed. Mol Cell. 2015;58:870–85.
Cui L, Mirza AH, Zhang S, Liang B, Liu P. Lipid droplets and mitochondria are anchored during brown adipocyte differentiation. Protein Cell. 2019;10:921–6.
Monzel AS, Enríquez JA, Picard M. Multifaceted mitochondria: moving mitochondrial science beyond function and dysfunction. Nat Metab. 2023;5:546–62.
Cicuéndez B, Mora A, López JA, Curtabbi A, Pérez-García J, Porteiro B, et al. Absence of MCJ/DnaJC15 promotes brown adipose tissue thermogenesis. Nat Commun. 2025;16:229.
Moore TM, Cheng L, Wolf DM, Ngo J, Segawa M, Zhu X, et al. Parkin regulates adiposity by coordinating mitophagy with mitochondrial biogenesis in white adipocytes. Nat Commun. 2022;13:6661.
Chen S, Yuan W, Huang Q, Xiong X, Wang C, Zeng W, et al. Asprosin contributes to pathogenesis of obesity by adipocyte mitophagy induction to inhibit white adipose browning in mice. Int J Obes. 2024;48:913–22.
Mottillo EP, Desjardins EM, Crane JD, Smith BK, Green AE, Ducommun S, et al. Lack of adipocyte AMPK exacerbates insulin resistance and hepatic steatosis through brown and beige adipose tissue function. Cell Metab. 2016;24:118–29.
Feng Z, Zhang X, Zhou J, Li Q, Chu L, Di G, et al. An in vitro-transcribed circular RNA targets the mitochondrial inner membrane cardiolipin to ablate EIF4G2+/PTBP1+ pan-adenocarcinoma. Nat Cancer. 2024;5:30–46.
Wang H, Liang Y, Zhang T, Yu X, Song X, Chen Y, et al. C-IGF1R encoded by cIGF1R acts as a molecular switch to restrict mitophagy of drug-tolerant persister tumour cells in non-small cell lung cancer. Cell Death Differ. 2023;30:2365–81.
Xie L, Huang W, Fang Z, Ding F, Zou F, Ma X, et al. CircERCC2 ameliorated intervertebral disc degeneration by regulating mitophagy and apoptosis through miR-182-5p/SIRT1 axis. Cell Death Dis. 2019;10:1–13.
Zhao N, Liu D, Song H, Zhang X, Yan C, Han Y. Identification of critical endoplasmic reticulum stress-related genes in advanced atherosclerotic plaque. Sci Rep. 2025;15:2107.
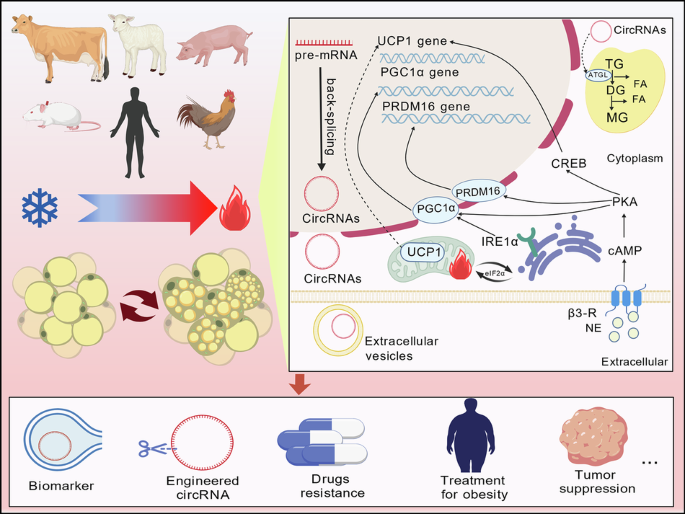
Chen Y, Wu Z, Huang S, Wang X, He S, Liu L, et al. Adipocyte IRE1α promotes PGC1α mRNA decay and restrains adaptive thermogenesis. Nat Metab. 2022;4:1166–84.
Cao Z, Xiao Q, Dai X, Zhou Z, Jiang R, Cheng Y, et al. circHIPK2-mediated σ-1R promotes endoplasmic reticulum stress in human pulmonary fibroblasts exposed to silica. Cell Death Dis. 2017;8:1–13.
Chakrabarty Y, Yang Z, Chen H, Chan DC. The HRI branch of the integrated stress response selectively triggers mitophagy. Mol Cell. 2024;84:1090–1100.e6.
Zhao Q, Liu J, Deng H, Ma R, Liao JY, Liang H, et al. Targeting mitochondria-located circRNA SCAR alleviates NASH via reducing mROS output. Cell. 2020;183:76–93.e22.
Chen C, Zhang X, Deng Y, Cui Q, Zhu J, Ren H, et al. Regulatory roles of circRNAs in adipogenesis and lipid metabolism: emerging insights into lipid-related diseases. FEBS J. 2021;288:3663–82.
Liu Y, Yang Y, Xu C, Liu J, Chen J, Li G, et al. Circular RNA circGlis3 protects against islet β-cell dysfunction and apoptosis in obesity. Nat Commun. 2023;14:351.
Malviya A, Bhuyan R. The recent advancements in circRNA research: From biogenesis to therapeutic interventions. Pathol Res Pr. 2023;248:154697.
Latreille M, Hausser J, Stützer I, Zhang Q, Hastoy B, Gargani S, et al. MicroRNA-7a regulates pancreatic β cell function. J Clin Invest. 2014;124:2722–35.
Gawrieh S, Opara EC, Koch TR. Oxidative stress in nonalcoholic fatty liver disease: pathogenesis and antioxidant therapies. J Investig Med Publ Am Fed Clin Res. 2004;52:506–14.
Chen X, Tan QQ, Tan XR, Li SJ, Zhang XX. Circ_0057558 promotes nonalcoholic fatty liver disease by regulating ROCK1/AMPK signaling through targeting miR-206. Cell Death Dis. 2021;12:1–12.
Tang T, Zhou J, Wang M, Xia S, Sun W, Jia X, et al. Transcriptomics reveals the regulatory mechanisms of circRNA in the muscle tissue of cows with ketosis postpartum. Genomics. 2025;117:111008.
Nguyen HP, An K, Ito Y, Kharbikar BN, Sheng R, Paredes B, et al. Implantation of engineered adipocytes suppresses tumor progression in cancer models. Nat Biotechnol. 2025;1–17.
Fei D, Wang F, Wang Y, Chen J, Chen S, Fan L, et al. Circular RNA ACVR2A promotes the progression of hepatocellular carcinoma through mir-511-5p targeting PI3K-Akt signaling pathway. Mol Cancer. 2024;23:159.
Li Z, Yanfang W, Li J, Jiang P, Peng T, Chen K, et al. Tumor-released exosomal circular RNA PDE8A promotes invasive growth via the miR-338/MACC1/MET pathway in pancreatic cancer. Cancer Lett. 2018;432:237–50.
Wang T, He M, Zhang X, Guo Z, Wang P, Long F. Deciphering the impact of circRNA-mediated autophagy on tumor therapeutic resistance: a novel perspective. Cell Mol Biol Lett. 2024;29:60.
Wong RJ. Epidemiology of metabolic dysfunction-associated steatotic liver disease (MASLD) and alcohol-related liver disease (ALD). Metab Target Organ Damage. 2024;4:35.
Llamoza-Torres CJ, Fuentes-Pardo M, Ramos-Molina B. Metabolic dysfunction-associated steatotic liver disease: a key factor in hepatocellular carcinoma therapy response. Metab Target Organ Damage. 2024;4:40.
Zheng JD, He TL, Chen JX, Zhao JX, Zhang SB, Yang ZG. Decoding the functional network of circular RNAs encoding proteins in hepatocellular carcinoma: from carcinogenesis to clinical transformation. J Adv Res. 2025;S2090-1232(25)00700-3.
Zeng Q, Liu CH, Wu D, Jiang W, Zhang N, Tang H. LncRNA and circRNA in patients with non-alcoholic fatty liver disease: a systematic review. Biomolecules. 2023;13:560.
Hu Y, Cai ZR, Huang RZ, Wang DS, Ju HQ, Chen DL. Circular RNA circPHLPP2 promotes tumor growth and anti-PD-1 resistance through binding ILF3 to regulate IL36γ transcription in colorectal cancer. Mol Cancer. 2024;23:272.
Wang X, Zhang C, Song H, Yuan J, Zhang L, He J. CircCCDC66: emerging roles and potential clinical values in malignant tumors. Front Oncol. 2022;12:1061007.
Nie J, Yang R, Zhou R, Deng Y, Li D, Gou D, et al. Circular RNA circFARSA promotes the tumorigenesis of non-small cell lung cancer by elevating B7H3 via sponging miR-15a-5p. Cell Cycle Georget Tex. 2022;21:2575–89.
Hafez AK, Zimmerman AJ, Papageorgiou G, Chandrasekaran J, Amoah SK, Lin R, et al. A bidirectional competitive interaction between circHomer1 and Homer1b within the orbitofrontal cortex regulates reversal learning. Cell Rep. 2022;38:110282.
Moon J, Kim D, Kim EK, Lee SY, Na HS, Kim GN, et al. Brown adipose tissue ameliorates autoimmune arthritis via inhibition of Th17 cells. Sci Rep. 2020;10:12374.
Rodríguez A, Becerril S, Hernández-Pardos AW, Frühbeck G. Adipose tissue depot differences in adipokines and effects on skeletal and cardiac muscle. Curr Opin Pharm. 2020;52:1–8.
Quesada-López T, Cereijo R, Turatsinze JV, Planavila A, Cairó M, Gavaldà-Navarro A, et al. The lipid sensor GPR120 promotes brown fat activation and FGF21 release from adipocytes. Nat Commun. 2016;7:13479.
Li Y, Zheng F, Xiao X, Xie F, Tao D, Huang C, et al. CircHIPK3 sponges miR-558 to suppress heparanase expression in bladder cancer cells. EMBO Rep. 2017;18:1646–59.
Singh K, Sethi P, Datta S, Chaudhary JS, Kumar S, Jain D, et al. Advances in gene therapy approaches targeting neuro-inflammation in neurodegenerative diseases. Ageing Res Rev. 2024;98:102321.
Ma N, Pan J, Ye X, Yu B, Zhang W, Wan J. Whole-transcriptome analysis of APP/PS1 mouse brain and identification of circRNA-miRNA-mRNA networks to investigate AD pathogenesis. Mol Ther Nucleic Acids. 2019;18:1049–62.
Dolinar A, Koritnik B, Glavač D, Ravnik-Glavač M. Circular RNAs as potential blood biomarkers in amyotrophic lateral sclerosis. Mol Neurobiol. 2019;56:8052–62.
Wang S, Zhang K, Tan S, Xin J, Yuan Q, Xu H, et al. Circular RNAs in body fluids as cancer biomarkers: the new frontier of liquid biopsies. Mol Cancer. 2021;20:13.
Guo X, Tan W, Wang C. The emerging roles of exosomal circRNAs in diseases. Clin Transl Oncol Publ Fed Span Oncol Soc Natl Cancer Inst Mex. 2021;23:1020–33.
Yu L, Wang C, Zhang D, Liu M, Liu T, Pan B, et al. Exosomal circ_0008285 in follicle fluid regulates the lipid metabolism through the miR-4644/ LDLR axis in polycystic ovary syndrome. J Ovarian Res. 2023;16:113.
Zhu Z, Huang J, Li X, Xing J, Chen Q, Liu R, et al. Gut microbiota regulate tumor metastasis via circRNA/miRNA networks. Gut Microbes. 2020;12:1788891.
He TL, Chen QY, Li HF, Mao JN, Luo J, Ma DJ, et al. The potential mechanism of MicroRNA involvement in the regulation of muscle development in weaned piglets by tryptophan and its metabolites. BMC Genomics. 2025;26:330–44.
He TL, Ma JY, Liu S, Ma BY, You JT, Wang JJ, et al. MicroRNA-microbiota interactions: Emerging strategies for modulating intestinal homeostasis and enhancing host health. iMetaOmics. 2025;2:e57.
Zhen XJ, Hu RT, Liu NN, Dou JF, Wu T, Zhang YL, et al. CircRNA-mediated ceRNA regulatory networks: transcriptomic insights into obesity type 2 diabetes progression and treatment strategies. Diabetol Metab Syndr. 2025;17:57.
Jiang B, Zhang J, Sun X, Yang C, Cheng G, Xu M, et al. Circulating exosomal hsa_circRNA_0039480 is highly expressed in gestational diabetes mellitus and may be served as a biomarker for early diagnosis of GDM. J Transl Med. 2022;20.
Farinas A, Rutledge J, Bot VA, Western D, Ying K, Lawrence KA, et al. Disruption of the cerebrospinal fluid–plasma protein balance in cognitive impairment and aging. Nat Med. 2025;1–12.
Tian L, Gao Y, Zi L, Zhe R, Yang J. Dual Cas12a and multiplex crRNA CRISPR strategy ultrasensitive detection novel circRNA biomarker for the diagnosis of ovarian cancer. BMC Cancer. 2025;25:695.
Liu X, Zhang Y, Zhou S, Dain L, Mei L, Zhu G. Circular RNA: An emerging frontier in RNA therapeutic targets, RNA therapeutics, and mRNA vaccines. J Control Release J Control Release Soc. 2022;348:84–94.
Bai Y, Liu D, He Q, Liu J, Mao Q, Liang Z. Research progress on circular RNA vaccines. Front Immunol. 2022;13:1091797.
He TL, Li CL, Chen QY, Li RQ, Luo J, Mao JN, et al. Combined analysis of lncRNA and mRNA emphasizes the potential role of tryptophan-mediated regulation of muscle development in weaned piglets by lncRNA. J Anim Sci. 2024;102:skae264.
Chen J, Gao Y, Zhong J, Wu X, Leng Z, Liu M, et al. Lnc-H19-derived protein shapes the immunosuppressive microenvironment of glioblastoma. Cell Rep Med. 2024;5:101806.
Zhang L, Liang D, Chen C, Wang Y, Amu G, Yang J, et al. Circular siRNAs for reducing off-target effects and enhancing long-term gene silencing in cells and mice. Mol Ther Nucleic Acids. 2018;10:237–44.
Liang J, Shao W, Ni P, Liu Q, Kong W, Shen W, et al. siRNA/CS-PLGA nanoparticle system targeting knockdown intestinal SOAT2 reduced intestinal lipid uptake and alleviated obesity. Adv Sci. 2024;11:2403442.
Hou X, Zaks T, Langer R, Dong Y. Lipid nanoparticles for mRNA delivery. Nat Rev Mater. 2021;6:1078–94.
Zhang Z, Ma B, Li B, Li Z, Gao M, Zhao H, et al. Cardiolipin-mimic lipid nanoparticles without antibody modification delivered senolytic in vivo CAR-T therapy for inflamm-aging. Cell Rep Med. 2025;6:102209.
Huang K, Li N, Li Y, Zhu J, Fan Q, Yang J, et al. Circular mRNA vaccine against SARS-COV-2 variants enabled by degradable lipid nanoparticles. ACS Appl Mater Interfaces. 2025;17:4699–710.
Zhao Z, Shan X, Ding J, Ma B, Li B, Huang W, et al. Boosting RNA nanotherapeutics with V-ATPase activating non-inflammatory lipid nanoparticles to treat chronic lung injury. Nat Commun. 2025;16:6477.
Bitounis D, Jacquinet E, Rogers MA, Amiji MM. Strategies to reduce the risks of mRNA drug and vaccine toxicity. Nat Rev Drug Discov. 2024;23:281–300.
Fan Q, Yang L, Zhang X, Peng X, Wei S, Su D, et al. The emerging role of exosome-derived non-coding RNAs in cancer biology. Cancer Lett. 2018;414:107–15.
Maretty L, Gill D, Simonsen L, Soh K, Zagkos L, Galanakis M, et al. Proteomic changes upon treatment with semaglutide in individuals with obesity. Nat Med. 2025;31:267–77.
Carrasco-Zanini J, Pietzner M, Davitte J, Surendran P, Croteau-Chonka DC, Robins C, et al. Proteomic signatures improve risk prediction for common and rare diseases. Nat Med. 2024;30:2489–98.
Jia X, He X, Huang C, Li J, Dong Z, Liu K. Protein translation: biological processes and therapeutic strategies for human diseases. Signal Transduct Target Ther. 2024;9:44.
Effiong UM, Khairandish H, Ramirez-Velez I, Wang Y, Belardi B. Turn-on protein switches for controlling actin binding in cells. BioRxiv Prepr Serv Biol. 2023;2023.10.26.561921.
Xu T, Wang M, Jiang L, Ma L, Wan L, Chen Q, et al. CircRNAs in anticancer drug resistance: recent advances and future potential. Mol Cancer. 2020;19:127.
Song S, Shi Y, Zeng D, Xu J, Yang Y, Guo W, et al. circANKRD28 inhibits cisplatin resistance in non-small-cell lung cancer through the miR-221-3p/SOCS3 axis. J Gene Med. 2023;25:e3478.
Liang R, He Z, Zhao KT, Zhu H, Hu J, Liu G, et al. Prime editing using CRISPR-Cas12a and circular RNAs in human cells. Nat Biotechnol. 2024;42:1867–75.
Kordyś M, Sen R, Warkocki Z. Applications of the versatile CRISPR-Cas13 RNA targeting system. Wiley Interdiscip Rev RNA. 2022;13:e1694.
Tong M, Palmer N, Dailamy A, Kumar A, Khaliq H, Han S, et al. Robust genome and cell engineering via in vitro and in situ circularized RNAs. Nat Biomed Eng. 2025;9:109–26.
Wang Z, Lei X. Matrix factorization with neural network for predicting circRNA-RBP interactions. BMC Bioinforma. 2020;21:229.
Chiang TW, Mai TL, Chuang TJ. CircMiMi: a stand-alone software for constructing circular RNA-microRNA-mRNA interactions across species. BMC Bioinforma. 2022;23:164.
Chaabane M, Williams RM, Stephens AT, Park JW. circDeep: deep learning approach for circular RNA classification from other long non-coding RNA. Bioinformatics. 2020;36:73–80.
Aufiero S, Reckman YJ, Pinto YM, Creemers EE. Circular RNAs open a new chapter in cardiovascular biology. Nat Rev Cardiol. 2019;16:503–14.
Liang D, Li G. Pulling the trigger: noncoding RNAs in white adipose tissue browning. Rev Endocr Metab Disord. 2024;25:399–420.
Sleiman L, Dinescu S. Role of non-coding RNAs in white and brown adipose tissue differentiation and development. Non-Coding RNA. 2025;11:30.
Dong X, Zhou Z, Wang Y, Nisar A, Pu S, Lv L, et al. CircAge: a comprehensive resource for aging-associated circular RNAs across species and tissues. Genomics Proteomics Bioinformatics. 2025;qzaf044.
Zeng W, Zheng L, Li Y, Yang J, Mao T, Zhang J, et al. Engineered extracellular vesicles for delivering functional Cas9/gRNA to eliminate hepatitis B virus cccDNA and integration. Emerg Microbes Infect. 2024;13:2284286.
Ruotsalainen AK, Kettunen S, Suoranta T, Kaikkonen MU, Ylä-Herttuala S, Aherrahrou R. The mechanisms of Chr.9p21.3 risk locus in coronary artery disease: where are we today?. Am J Physiol Heart Circ Physiol. 2025;328:H196–208.
Wu W, Zhang J, Cao X, Cai Z, Zhao F. Exploring the cellular landscape of circular RNAs using full-length single-cell RNA sequencing. Nat Commun. 2022;13:3242.
Zhang L, Li X, Gao H, Chang W, Li P. Gut microbiota-lncRNA/circRNA crosstalk: implications for different diseases. Crit Rev Microbiol. 2024;1–15.
Hu C, Feng X, Ma Y, Wei D, Zhang L, Wang S, et al. CircADAMTS16 inhibits differentiation and promotes proliferation of bovine adipocytes by targeting miR-10167-3p. Cells. 2023;12:1175.
Chen S, Song P, Wang Y, Wang Z, Xue J, Jiang Y, et al. CircMAPK9 promotes adipogenesis through modulating hsa-miR-1322/FTO axis in obesity. iScience. 2023;26:107756.
Kang Z, Zhang S, Jiang E, Wang X, Wang Z, Chen H, et al. circFLT1 and lncCCPG1 Sponges miR-93 to Regulate the Proliferation and Differentiation of Adipocytes by Promoting lncSLC30A9 Expression. Mol Ther Nucleic Acids. 2020;22:484–99.
Ding Z, Sun D, Han J, Shen L, Yang F, Sah S, et al. Novel noncoding RNA CircPTK2 regulates lipolysis and adipogenesis in cachexia. Mol Metab. 2021;53:101310.
Yue X, Fan M, Liang Y, Qiao L, Liu J, Pan Y, et al. circITGB1 Regulates Adipocyte Proliferation and Differentiation via the miR-23a/ARRB1 Pathway. Int J Mol Sci. 2023;24:1976.
Ma Z, Chen Y, Qiu J, Guo R, Cai K, Zheng Y, et al. CircBTBD7 inhibits adipogenesis via the miR-183/SMAD4 axis. Int J Biol Macromol. 2023;253:126740.
Rong X, Li R, Gong T, Li H, Zhao X, Cao G, et al. CircMEF2C(2, 3) modulates proliferation and adipogenesis of porcine intramuscular preadipocytes by miR-383/671-3p/MEF2C axis. iScience. 2024;27:109710.
Source link