Industries | The Big Take
Lisa Harding’s first seven patients are already getting high by 8 a.m.
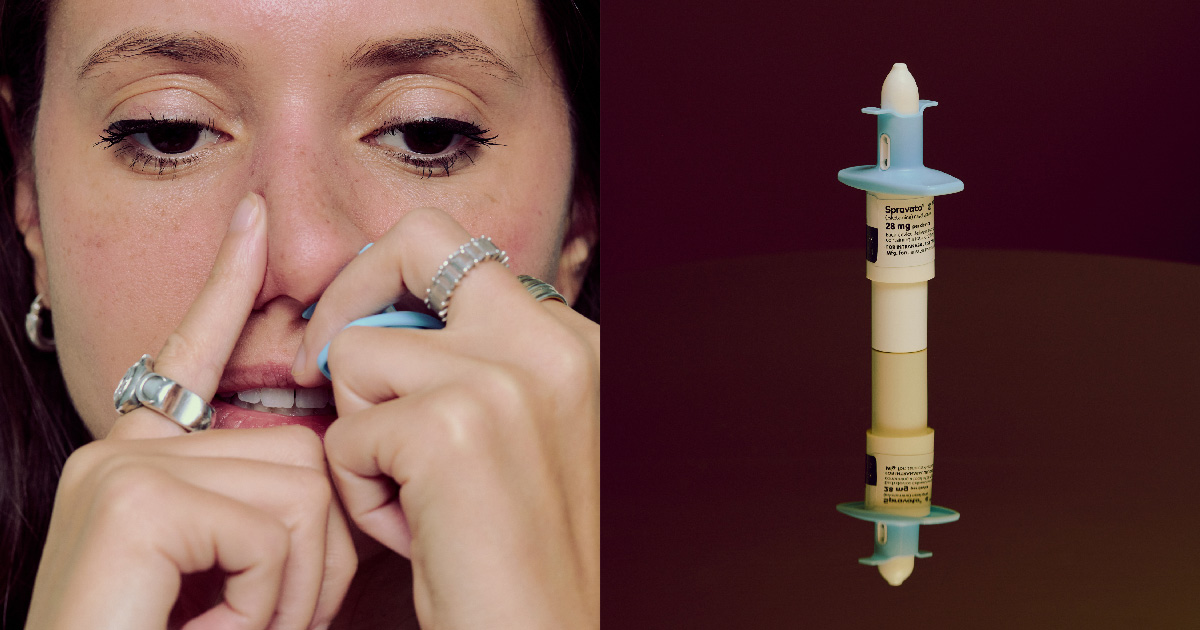
With a few spritzes, Johnson & Johnson’s ketamine-derived nasal spray Spravato is rewiring their brains. Their reactions vary: Some patients sob uncontrollably; others close their eyes and drift off. A few reach for J&J-designed coloring books.
By 10 a.m., those patients are out the door. Fifteen minutes later, seven more take their seats at the Milford, Connecticut, clinic.
They’re all chasing what some describe as a life-changing reset after struggling with depression. Dylan Field Turner, 54, had cycled through antidepressants for a decade before trying Spravato. The drug is esketamine, which is chemically related to ketamine but more potent in smaller doses, and can cause some psychedelic-like effects. “It shakes things loose,” he said. “It’s like changing your spark plugs.”
With the help of doctors such as Harding, J&J has turned Spravato into a $1.7 billion blockbuster. Clinics like hers took what’s essentially a psychedelic-like trip and fit it into the buttoned-up world of psychiatric medicine and insurance claims. Harding designed her clinic, the Mood Institute, not only to meet strict federal guidelines around monitoring, administering and storing the drug, but also to handle enough patients to run a sustainable practice.
Now, J&J is paying her to teach others around the world about Spravato, turning the drug launch into a template for a new kind of psychiatric care. Despite the much-hyped potential of psychedelics as mental health treatments — particularly as they’ve become more acceptable in mainstream culture — regulatory setbacks and scientific hurdles have kept the market from taking off. Spravato is the first drug in the arena to figure it out.
If more psychedelic-adjacent therapies win approval, analysts estimate the market could generate roughly $7 billion in annual sales by 2032. Those approvals could come sooner than originally expected. On April 18, President Donald Trump signed an executive order directing his administration to expand access to psychedelic treatments. Days later, US drug regulators awarded national priority vouchers to three companies developing such therapies in an effort to speed up the review process.
When they arrive, the next generation of psychedelics “will be able to seamlessly fit into the existing Spravato-administering interventional psychiatric centers,” said RBC Capital Markets analyst Brian Abrahams.
“We’re doing really well with Spravato,” Chief Executive Officer Joaquin Duato told investors on a call last year. “Everybody’s trying to copy our model,” he said, referring to the infrastructure the company has built around the drug.
Off the success of Spravato, neurology has become one of J&J’s top investment priorities. Last year it spent $14.6 billion on Intra-Cellular Therapies Inc., which makes Caplyta for major depressive disorder, and scooped up another experimental medicine that’s being studied for Alzheimer’s-related agitation and anxiety. But it remains to be seen whether J&J will wade further into psychedelics even as rivals like AbbVie Inc. are swooping in with their own novel depression drugs.
More than 332 million people worldwide live with depression — and about a third of adults will not respond to oral antidepressants alone.
When Spravato first launched in 2019, executives and analysts had high hopes. It was the first major breakthrough in depression since Prozac and the first psychedelic-adjacent drug to get US Food and Drug Administration approval to treat a mental health condition.
But J&J soon realized that the psychiatry system couldn’t accommodate a treatment that requires patients to trip under supervision for hours. And to make matters worse, Spravato’s launch coincided with the Covid-19 pandemic, when in-person care ground to a screeching halt.
Most insurers paid for Spravato in the same way they paid for traditional office visits, not as a time-intensive process. Psychiatrists’ offices, where people tend to seek treatment for depression, weren’t set up for something more akin to a medical procedure, nor did they have the staff to oversee it. They needed to get certified and maintain airtight documentation to withstand federal audits. Even if they checked all the requisite boxes, it was unclear whether insurers would cover the hours-long patient visits.
Spravato sales were so low for the first two years that J&J didn’t break them out on its balance sheet. The rollout was “very slow and disappointing,” said Vamil Divan, a senior research analyst at Guggenheim Partners, particularly after the company had been hyping the drug for at least a couple of years prior.
J&J said the initial slowness was to be expected. “New models of care often take time to see significant uptake,” said Jane Tiller, J&J’s global head of neuroscience clinical development.
Spravato Sales Soar
J&J’s depression treatment is now a key growth driver after early stumbles
Some doctors, however, felt misled by the company’s early messaging around the drug. “I was angry at J&J for a long time,” Harding said. “They kept selling this pipe dream to every other clinician that you should open a thing and do it yourself, without understanding that the mathematics were upside down.”
Even inside the company, there were naysayers who weren’t sold on the Spravato pitch, said Husseini Manji, J&J’s former head of neuroscience, who developed and helped launch the drug. “Traditionalists” were skeptical that such a tightly controlled, doctor-administered drug could take off, he said. That’s why for the initial launch the drugmaker targeted big university hospitals that were better set up to manage Spravato’s strict requirements — and potential risk of abuse.
“I deliberately wanted to do it slowly, to start with academic centers because I didn’t want anything to go wrong,” Manji said.
UC San Diego signed up to administer Spravato as soon as it was approved. But in its first year, it only had three patients taking the drug.
The logistics were crushing. Each session tied up clinic rooms for hours and generated stacks of paperwork for the FDA, the Drug Enforcement Administration and insurers — all of whom could audit the program at any time. Staff had to track every dose from supplier to patient. If they didn’t use it within 14 days, they had to toss it.
“It was really, really difficult,” said Ashraf Elmashat, then an associate clinical professor in UC San Diego’s psychiatry department and medical director of specialty clinics. The team struggled to navigate the drug’s strict requirements as J&J was still in the early days of providing guidance to clinics, he said.
Insurance approvals dragged on for months, specialty pharmacies often didn’t stock the drug and buying it directly meant risking thousands of dollars if reimbursement fell through. Meanwhile, few physicians — let alone patients — were familiar with the treatment. Within a year of the rollout, UC San Diego temporarily shelved the program.
On a 2021 earnings call, an analyst questioned whether enthusiasm for the drug had cooled inside the company. J&J insisted “its excitement has not diminished at all.” When the drug came up on subsequent calls, the company blamed the weak sales on the Covid-19 pandemic and the “conservative” nature of psychiatrists.
If big hospitals were having a rough go at managing Spravato, it was all but impossible for smaller psychiatry clinics — where most people seek treatment for depression — to make it work.
One of the main issues was billing, which Manji said he underestimated. “I thought, OK, it’s a no-brainer,” he said. But it took years to untangle post-launch.
“If Spravato hadn’t started working for me… it’s possible I would not have made it”
A company spokesperson said J&J expanded its access to educational resources for clinics but declined to elaborate further on how it managed the issue.
To hear clinic owners tell it, the company pulled seemingly every lever it could. Billing codes didn’t exist for the recovery time or the administration of Spravato, only for a standard office visit. J&J worked with clinics and insurers to create those — a crucial shift that made the business model viable. The company also provides rebates to support the increasing number of patients with high-deductible insurance plans.
At the University of Southern California’s Keck Medicine, J&J representatives met repeatedly with its leaders to walk them through the mechanics of launching and running a Spravato program, including how to meet the numerous documentation requirements, according to Steven Siegel, vice president for behavioral health at the school. J&J even hosted dinners where doctors could swap notes and share advice. Guidance from the company helped Keck make its Spravato operation more sustainable and ensured it was getting paid more than “$2 to administer several hundred dollars worth of time,” he said.
Keck currently treats 11 patients in its program, which now breaks even thanks to insurance reimbursement, while its IV ketamine treatments cost the clinic money.
As the math started to make more sense at more facilities, some clinics figured out how to turn Spravato into a lucrative business.
It can be “extraordinarily profitable,” said Jay Godfrey, who runs Nushama Wellness. The Manhattan clinic treats as many as 40 Spravato patients a day in a space that looks more like a nature-themed spa than a sterile medical office. He learned early on that some insurance plans let clinics act as the pharmacy and buy the drug in bulk. Doing so can lift revenue by as much as 35% per patient, Godfrey said.
Spravato is now about half of Nushama’s business, and often brings in more profit than its other main therapy, IV ketamine. He now operates two clinics in New York and opened a third in the fall in Miami.
Clinics also found success by speaking to patients in new and more direct ways. InnerMost, a Manhattan center that combines Spravato with talk therapy, designed its space to be comforting and homey, a decision its co-founder Casey Paleos said was meant to signal the experience will feel safe and supportive. It also, like many ketamine clinics, advertises on Instagram.
That’s how many people hear of these drugs, Harding said.
That was the case for Mike A. Bryan. The 46-year-old had struggled with depression, mania and bipolar disorder for more than a decade. In 2023, while scrolling Instagram, he saw an ad for Greenbrook TMS in the suburbs of St. Louis, Missouri. Unlike IV ketamine, his Medicaid plan covered the treatment. After his first dose in January 2024, the suicidal thoughts that once dominated his life began to taper, he said.
“I can’t say a hundred percent that I would not be here today if Spravato hadn’t started working for me,” Bryan said. “But it’s possible – it’s possible I would not have made it.”
J&J itself spent more than $3.2 million on television ads in 2024, according to iSpot.tv, promising viewers they “may begin to feel like yourself again.” Sales continue to rise despite the drugmaker not keeping up the ad blitz. Other companies have swooped in with their own campaigns. Keta Medical Center in the Northeast paid for a New York City subway banner inviting riders to “Shpritz Your Depression Away!”
The messaging gives some physicians pause. The hype and patient testimonies can make it seem like these new drugs are a cure-all for people with all sorts of psychiatric conditions, said Jerrold Rosenbaum, professor of psychiatry at Harvard Medical School.
It can create “a lot of enthusiasm and optimism, which is probably true for a subset of patients,” he said. But not everyone will have the same experience or see the same benefit, he warned.
Other doctors, while acknowledging the promise of Spravato, still have questions about its effectiveness. Susan Conroy, assistant professor of psychiatry at Indiana University School of Medicine, said it’s not always clear whether people feel better because they’re under the influence of the drug, or because the drug is treating their underlying symptoms. “That is a really hard thing to tease out,” she said.
J&J said the drug works, in part, by increasing the levels of glutamate in the brain, which supports improvements that go beyond same-day effects of the treatment. “Spravato is designed to treat the underlying neurobiology of treatment-resistant depression rather than just masking symptoms,” a spokesperson said.
Still, demand shows no signs of slowing. The number of sites certified to administer Spravato has jumped from 2,800 in 2024 to more than 7,000 in 2026. Sales have more than doubled since 2023, and Wall Street expects the drug to generate about $2.3 billion for J&J this year, especially as it pushes into more international markets. The company has said those forecasts are an underestimate, said John Murphy, a senior industry analyst at Bloomberg Intelligence. In January 2025, the FDA expanded Spravato’s label after the company demonstrated it worked without background antidepressants — a move that further broadened its reach.
Rival drugmakers have taken note. Startups including Compass Pathways Plc, Definium Therapeutics Inc. and GH Research Plc are racing to enter the market for this novel class of drugs, while industry stalwart AbbVie recently made a billion-dollar bet on a next-generation psychedelic compound called bretisilocin.
It’s unclear how some of these bets will pay off. In the wake of the FDA’s 2024 rejection of an ecstasy-derived treatment from Lykos Therapeutics, companies were forced to re-evaluate their clinical trial designs and safety protocols, raising questions about the future of similar therapies. But analysts now say that case is unlikely to have any bearing on the FDA’s decisions around the broader psychedelic class of drugs — especially after Trump’s recent executive order.
“I’m pretty convinced that successful studies are going to generate approvals,” said RBC’s Abrahams.
With the systems Harding has in place now — including technology to help monitor patients and artificial intelligence software to make the process more efficient — she’s optimistic about managing the next generation of psychedelic-derived drugs.
Of course, challenges remain for the clinicians on the frontlines of treating patients. Insurance denials still occur, which means Harding has to use off-label IV ketamine when patients’ insurance plans don’t include Spravato.
“There’s still a lot of kinks to be worked out,” she said.
(Updates with data about prevalence of depression in 11th paragraph.)
Source link