In an evolving health landscape, emerging research continues to highlight concerns that could impact everyday wellbeing. Here’s the key update you should know about:
A data-driven tool built from nearly 200,000 individuals reveals who is most likely to develop serious obesity-related diseases, offering a smarter way to target treatment beyond BMI.
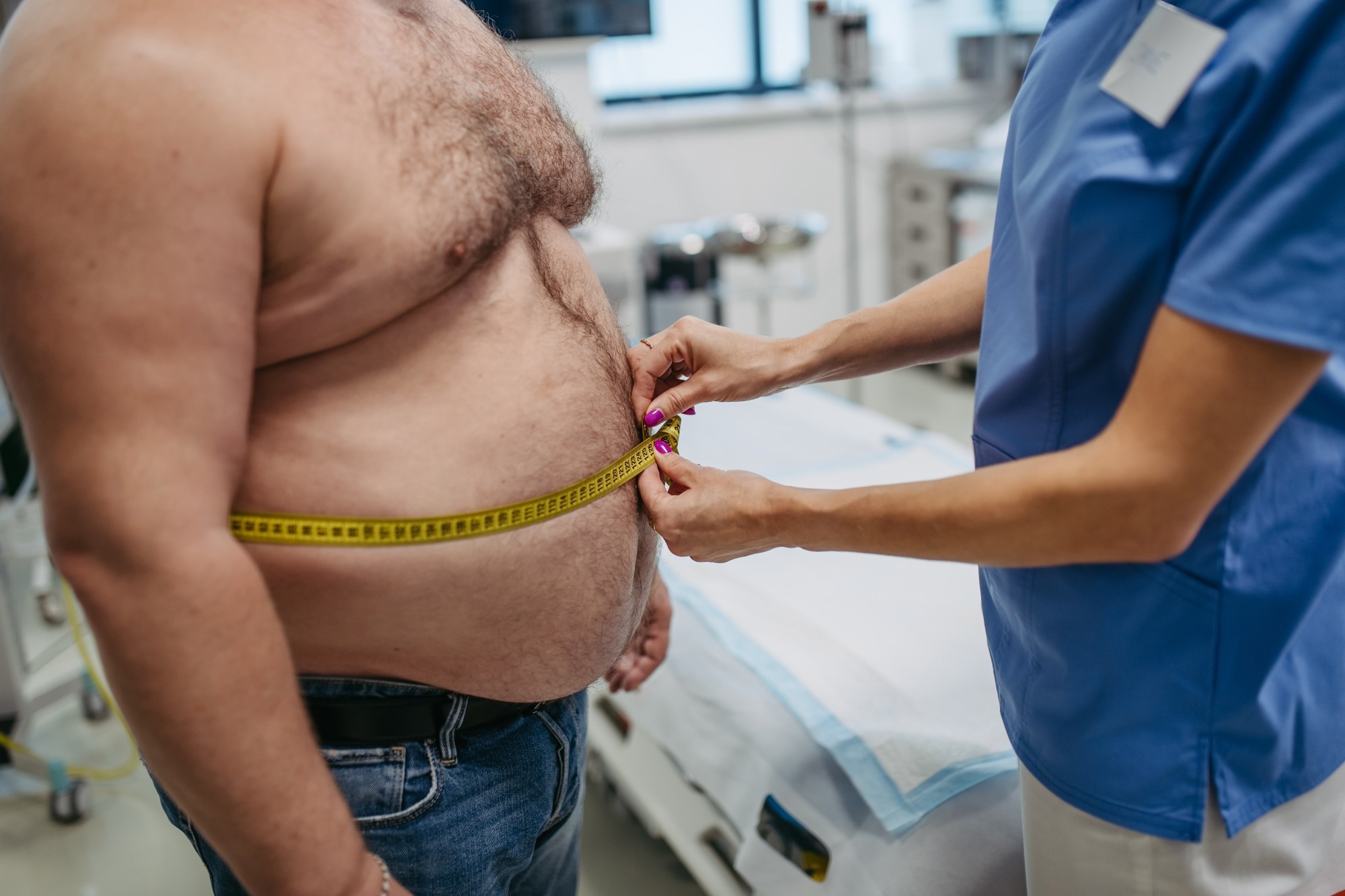
Study: Data-driven prioritization of high-risk individuals for weight loss interventions. Image credit: Halfpoint/Shutterstock.com
A recent Nature Medicine study developed and validated OBSCORE, a model that identifies obese or overweight individuals who are most likely to develop related health problems.
Obesity-Related Morbidity, Treatment Advances, and Resource Allocation
Obesity represents a chronic pathological condition that significantly increases susceptibility to metabolic diseases, including type 2 diabetes and cardiovascular disease, as well as mechanical complications such as sleep apnea. Collectively, these complications are responsible for approximately 5 million deaths and 130 million disability-adjusted life years annually, reflecting a threefold rise since 1990.
Emerging pharmacological agents, including semaglutide and tirzepatide, target the glucagon-like peptide-1 (GLP-1) and glucose-dependent insulinotropic polypeptide (GIP) pathways and have demonstrated substantial efficacy in managing obesity and its associated comorbidities. However, with 60–70 % of adults in Western populations classified as overweight or obese, the widespread implementation of pharmacotherapy presents considerable logistical and economic challenges for healthcare systems.
Efficient allocation of resources across pharmacological, dietary, behavioral, and surgical interventions requires precise identification and stratification of individuals at elevated risk for obesity-related complications. Sole reliance on body mass index (BMI) is inadequate. Current frameworks emphasize defining clinical obesity through a multidimensional lens that incorporates health status and phenotypic subtypes. Nevertheless, the specific parameters necessary for robust risk stratification remain unresolved, and a validated, data-driven framework for risk-based intervention is lacking.
Large-scale, contemporary population studies leveraging comprehensive baseline assessments and longitudinal healthcare record linkage now facilitate the integration of heterogeneous data modalities. While machine learning techniques can extract salient predictors from high-dimensional datasets, their ability to construct risk-based clinical tools for obesity management remains largely unexplored.
Identifying High-Risk Populations Through Predictive Modeling
The current study introduced OBSCORE, a risk prediction tool that identifies high-risk individuals for intervention. It employed the UK Biobank (UKB), a prospective, population-based study of about 500,000 UK residents aged 40–69.
The sample size was not predetermined statistically, and neither randomization nor blinding was used. To reflect clinical reality, exclusion criteria similar to those of the SURMOUNT-1 trial were used: adults with BMI ≥27 kg/m² were included with or without comorbidities. Those with contraindications or specific conditions were excluded.
A two-step machine learning framework combined LASSO for feature selection and a regularized Cox model for optimization and validation, focusing on the top 20 features for each complication. The cohort was split into feature selection, optimization, and validation sets, and model performance was assessed with bootstrapped C-indices.
To improve clinical utility, a single model with 20 core features was designed to potentially enable practical implementation in clinical settings by predicting multiple complications, and outcome-specific models were retrained as needed. A shared clinical model was externally validated in the European Prospective Investigation into Cancer (EPIC)-Norfolk cohort for a subset of outcomes and in the Genes & Health studies, which involved British Pakistani and Bangladeshi adults for type 2 diabetes.
OBSCORE Enables Precise, Individualized Risk Stratification Beyond BMI
Of nearly 500,000 UK Biobank participants, 197,264 met weight-loss trial criteria (97,421 overweight, 99,843 obese; median age 58). At baseline, 9.3 % had type 2 diabetes and 4.4 % had a major cardiac history. Over 10 years, 18 obesity-related complications were tracked, with arthropathy and hypertension being the most common, and 9,337 deaths were recorded. Of 2,390 candidate predictors, 2,078 remained after removing highly correlated variables.
General health, behavior, and clinical blood biomarkers gave the best predictive performance; polygenic scores performed worst. Including all features gave results similar to using just the top 20, except for diseases and medication, where using more features led to better performance.
To test prediction accuracy, different types of health data were added stepwise, using the top 20 features from each domain. Combining diagnoses, medications, and blood tests with general health information improved prediction accuracy, highlighting the value of clinical records. However, adding extra data like cardiopulmonary measures, body composition, metabolites, or genetic information provided little additional benefit for most outcomes.
The strongest predictive performance was observed for conditions like gout, chronic kidney disease, and type 2 diabetes. These results were largely driven by blood biomarkers; however, certain conditions, including diaphragmatic hernia, remained challenging to predict accurately.
Twenty shared, mostly uncorrelated clinical features were identified to create a single model, OBSCORE, that predicts risk across all 18 outcomes. This model prioritizes rapid, clinically available data over complex markers such as metabolites and genetics, which contributed little additional value.
OBSCORE demonstrated predictive accuracy comparable to more complex, outcome-specific models, with only minor differences. It consistently outperformed BMI-based approaches and models based on features from established risk scores (such as ASCVD and SCORE2), particularly for non-cardiovascular conditions like type 2 diabetes and sleep apnea. These findings were robust across patient subgroups and showed generalizability in external cohorts, highlighting the model’s potential applicability across populations.
When comparing detection rates across different false-positive thresholds, OBSCORE and the extended outcome-specific models performed similarly. Importantly, OBSCORE more effectively identified individuals at high risk, reflected by higher post-test probabilities for complications, especially for type 2 diabetes. While OBSCORE’s calibration was generally strong, occasional overestimation of risk was noted; this was mitigated through further statistical adjustment.
By stratifying individuals according to absolute risk, OBSCORE enables more targeted interventions. Notably, many people identified as high risk were overweight rather than obese, which exposes the limitations of relying solely on BMI for risk assessment. Risk also varied considerably within BMI categories, and OBSCORE offered much greater distinction between high- and low-risk groups than BMI alone, resulting in substantial differences in predicted outcomes. An interactive online tool was developed to allow users to explore personalized risk estimates.
In the SURMOUNT-1 trial, the medication tirzepatide reduced both body weight and waist-to-height ratio similarly across all OBSCORE risk groups. Those classified as higher risk saw, on average, greater absolute reductions, although relative reductions were slightly smaller. After treatment, OBSCORE risk estimates decreased significantly in all tirzepatide groups, indicating that the model is sensitive to changes in predicted risk following intervention.
OBSCORE offers scalable clinical decision-support potential
The current study developed and validated a risk prediction tool to better identify individuals at high risk of developing obesity-related complications. By accurately stratifying risk, this tool may help clinicians prioritize early intervention and allocate resources more efficiently. These findings support implementing data-driven, risk-centered approaches to personalize obesity management strategies as a complementary decision-support framework. Further validation, calibration, and definition of clinically meaningful risk thresholds are required before routine clinical adoption.
However, several limitations should be considered, including a focus on predominantly middle- to older-aged participants, the healthier-than-average UK Biobank population, potential biases from electronic health record–derived outcomes, and reduced predictive performance for certain conditions such as GERD and arthropathy. Additionally, the identified predictors are not necessarily causal and require further validation in diverse populations.