Mothers from across the country have spoken to Newsweek about their concern for their children after the Food and Drug Administration (FDA) has been rejecting various treatments for rare diseases that can be life-threatening.
One mother said she was “terrified” of losing access, as a result of an FDA rejection, to the drug her daughter relies on, while other mothers also said they were worried about how much longer they have left with their children.
The mother of a 15-year-old boy with Hunter Syndrome told Newsweek: “Every night when I put him in bed, I wonder if it’s the last time. Is this the last time I will see my boy alive, warm, and looking at me? It is always there.”
An FDA spokesperson told Newsweek: “The FDA is diligently committed to delivering cures and treatments to families in need in the safest way possible. That means requiring clear evidence of clinical benefit for the intended patient populations. Companies must demonstrate that benefit through strong data from a well-controlled and adequately run clinical trial.”
“The American people voted for an FDA that works for them—not just the sponsors—and the agency is delivering by refusing to rubber-stamp approvals. Under Commissioner Makary’s leadership, FDA approvals are at their peak, driven by the agency’s commitment to first-class science,” the spokesperson added.
The FDA’s Denials Of Rare Disease Treatments
Around 30 million Americans live with a rare disease, according to the FDA, and the agency’s commissioner himself said that, given the high number of Americans with a rare disease, “the problem isn’t rare—it’s a call to action.”
There are over 10,000 rare diseases, and around half of the U.S. population with a rare disease are children, according to the FDA. Many of these diseases are life-threatening, and most of them do not have approved treatments. Some patients with rare diseases gain access to potential treatments through clinical trials.
The FDA has announced a number of reforms designed to accelerate rare disease innovation including: common-sense flexibilities for manufacturing cell and gene therapies; embracing Bayesian statistics, which are better for small patient populations; for certain rare disease drug reviews, working with sponsors to complement traditional trial data with additional evidence such as evidence non-clinical models, pharmacodynamic data, or case reports; among other reforms.
However, according to Kim Higbee, a board member of the Hope For PDCD foundation, an organization supporting those with pyruvate dehydrogenase deficiency (PDCD), since the start of January 2025, the rare disease community has seen the FDA send 23 complete response letters regarding rare disease drugs.
The FDA will send out a complete response letter if it decides the drug in question cannot be approved in its present form. This typically means the drug is rejected or its approval is delayed until additional investigation into its safety and effectiveness as a treatment is completed.
The FDA’s rejections of rare disease treatments have prompted notable backlash in recent weeks, and Senator Ron Johnson has said he will be launching an investigation into the agency as a result.
For some children, the FDA’s lack of approval of certain treatments means that the drugs they have been relying on through medical trials may no longer be available to them, leaving their families to fear what that will mean for their futures.
PDCD also came into the spotlight recently when PGA golfer Akshay Bhatia said, after his win of the Arnold Palmer International tournament in Florida earlier this month, the victory was in honor of his niece, who had PDCD and passed away in December at age 6.
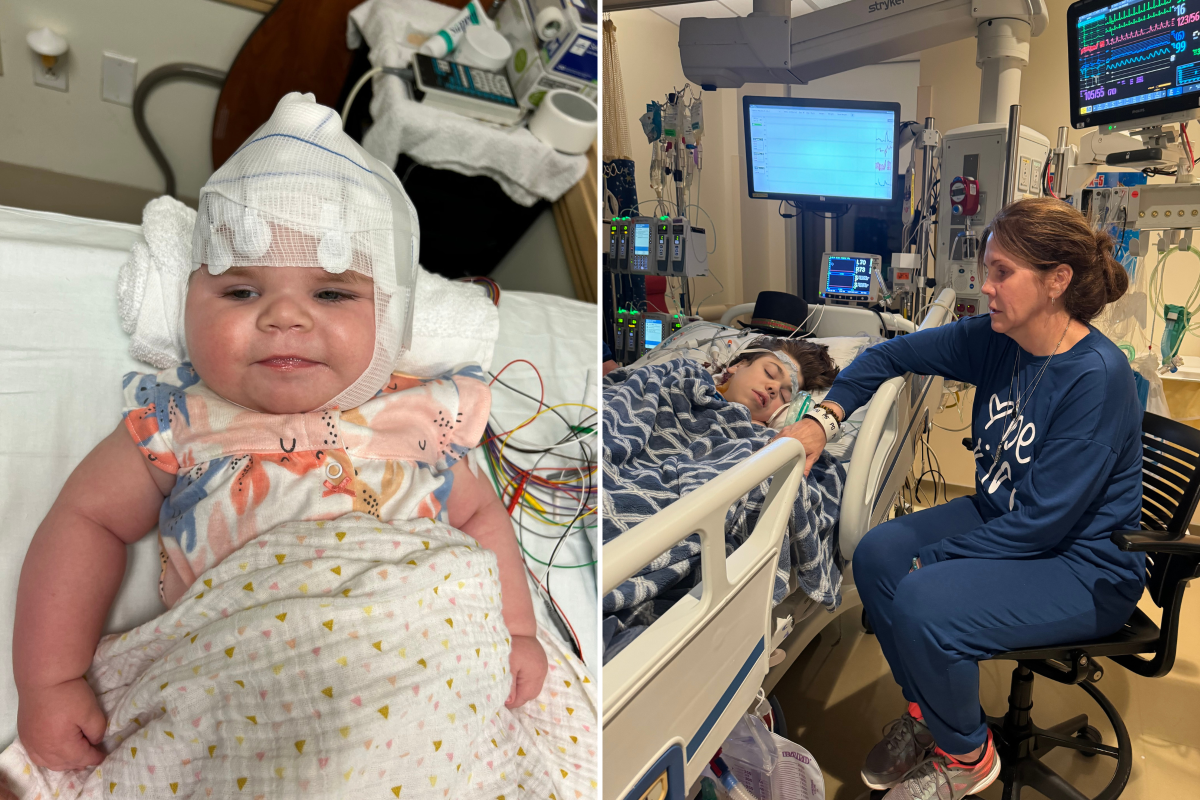
Meet 15-Year-Old Cole, Who Has Hunter Syndrome
One child affected by the FDA’s lack of approval of drug manufacturer Regenxbio’s Hunter syndrome gene therapy, RGX-121, last month, is 15-year-old Cole Stephens.
Cole’s mother, Kim, executive director of the Dr. Joseph Muenzer MPS Research and Treatment Center at the University of North Carolina at Chapel Hill, told Newsweek that he had a “fairly normal first two years of life,” but that certain symptoms prompted investigation with an ear, nose and throat (ENT) specialist.
The specialist then alerted Stephens that he thought Cole might have Hunter Syndrome. “In a few seconds, my dreams and expectations for my family, for Cole, were shattered. My baby boy was going to die,” Stephens told Newsweek.
After his diagnosis, there was around a two-year wait before Cole was able to enter a trial for enzyme replacement therapy (ERT). In that time, Stephens said she watched Cole closely for any changes to his condition, and slept next to him so she could monitor his breathing throughout the night.
At 15, Cole doesn’t speak, but he’s able to go for walks, feed himself and breathe without assistance. He said his last word at age 9, which was “Mommy.”
“Seeing your child deteriorate is the hardest thing you can imagine,” Stephens said. “After his diagnosis, I was watching every day for signs of disease progression. And then it started happening, and I was powerless to stop it. He was going to die, and I couldn’t help him.”
In its complete response letter, the FDA suggested that Regenxbio conduct a new study, treat additional patients, and conduct longer-term follow-up of the treatment going forward, delaying the trajectory of when the drug could be approved.
There is also another drug on the radar for Hunter Syndrome, tividenofusp alfa (or DNL310), but Stephens said that, as these drugs “slip away and get delayed, so does my hope.”
Stephens said she hoped to work with Johnson on his investigation into the FDA. “We have to break the pattern of rejection of rare disease treatments and the FDA’s refusal to use accelerated approval and biomarkers. The science is sound, and technicalities are killing our kids,” she said.
Meet 4-Year-Old Harlow, Who Has PDCD
Harlow, the 4-year-old daughter of Kim Higbee, has PDCD. Higbee told Newsweek that from birth, they were aware “something was wrong” with Harlow’s health, and that her diagnostic journey “started on day one.”
It was after a brain MRI came back abnormal when she was six months old that things progressed. The scan showed that one side of her brain was smaller than the other, and she was then referred quickly to genetics, where she was eventually diagnosed with PDCD at 9 months old.
“The older she got, the more we could see she wasn’t progressing like our first child had done,” Higbee said, who also has a daughter called Cassidy, aged 7.
When she was 10 months old, Harlow was able to get on the trial for Dichloroacetate (DCA), a potential treatment of PDCD, and Higbee said she’d been on an “open label” of the drug since April 2023—almost three years now.
Higbee said the combination of the treatment plus a medical ketogenic diet—a high-fat, very low-carbohydrate diet—had notable improvements on Harlow’s condition.
“You could see her awake windows extending longer, so she was more awake,” Higbee said, “you could see her starting to actually play with toys, before she wouldn’t be able to grasp anything, and starting to sit up.”
At 4, Harlow was able to crawl. “That was a big milestone for us, to get her mobility past just like the rolling mobility,” Higbee said. Harlow is also “pulling up to stand with assistance,” her mother added.
Harlow has to be fed with a feeding tube, so it can mean the morning school run is difficult for Higbee, as she has to prepare the ketogenic-appropriate food for Harlow, feed her with the tube which takes between 30 to 45 minutes, and then take her two daughters to two separate schools, as the one Cassidy goes to does not have the specific support in place for children with special needs.
While Higbee said she’s “grateful for how far [Harlow’s] come,” there are days when she wonders: “Why couldn’t it just be easy?”
When the FDA recently rejected Saol Therapeutics’ PDCD treatment drug—the one Harlow has been on for years—Higbee said she and the PDCD community felt “blindsided by it because we have seen our kids do things we never expected them to do.”
She added that it was “devastating,” and now leaves her with the “immediate fear” of whether Harlow will lose the drug she relies on. Given that the manufacturer will likely not be able to sustain the open-label access without FDA approval, it leaves the hopes of many families of children with the condition crushed.
“We’re fighting this as hard as we can,” Higbee said, explaining she’d traveled to Washington, D.C., a number of times already to speak to lawmakers. “Our families—rare disease families—should never have to fight the government while fighting for your child’s life.”
Meet 4-Year-Old Violet, Who Has PDCD
Another young girl living with PDCD is Violet, the 4-year-old daughter of Frances Pimentel. When Violet was born, Pimentel told Newsweek that there were “no warning signs” that she was not healthy. The family left the hospital following the birth “blissfully thinking our family was complete and life would continue as normal,” Pimentel said.
However, over time, Violet’s poor appetite and constant lethargy concerned Pimentel, who at the time worked in the infant feeding industry, and given her knowledge, sensed something was “seriously wrong.”
She tried to seek help from doctors, but she said she felt “continually gaslit and was told that I was overreacting.” It was when Violet was 8 months old that her concerns were finally taken seriously by a genetics team at the University of California, San Francisco. There, she was eventually diagnosed with PDCD. At the time, Violet could “not hold her head up, roll over, track toys, or even grasp and hold a toy,” Pimentel said.
Like Harlow, Violet was placed on a medical ketogenic diet and fed through a feeding tube, and later enrolled on the DCA clinical trial. “We thank God every day that we made that decision” to enroll her, Pimentel said.
Having been on the trail for three years, Violet’s progress has been “incredible to witness,” Pimentel said. She’s able to slightly widen what food she can eat, while making progress toward eating and drinking by mouth, walk short distances with assistance, and “we’ve lost count of how many words she can say.”
When Violet was first diagnosed, “I was devastated,” Pimentel said, but her grief led her to action, and she founded the parent-led patient advocacy organization, Hope For PDCD. “It has been therapeutic for me to channel my grief into advocacy for this condition,” she said, adding she was “also incredibly proud of my daughter and her determination.”
Following the FDA’s rejection of the drug Violet has been taking for years, Pimentel said she felt “fear and despair,” and was “terrified of losing access to the drug for Violet and all her PDCD brothers and sisters.”
“In the past year, rare disease advocates and biotech companies have experienced a devastating increase in denials and delays at the FDA, as well as an erratic and unpredictable application of the regulatory tools that Congress has given them,” she said. “For Violet, and for every child born with PDCD, we cannot afford to let delay become damage.”
In a polarized era, the center is dismissed as bland. At Newsweek, ours is different: The Courageous Center—it’s not “both sides,” it’s sharp, challenging and alive with ideas. We follow facts, not factions. If that sounds like the kind of journalism you want to see thrive, we need you.
When you become a Newsweek Member, you support a mission to keep the center strong and vibrant. Members enjoy: Ad-free browsing, exclusive content and editor conversations. Help keep the center courageous. Join today.
Source link